| CAS Number | 609-36-9 |
|---|---|
| Molecular Formula | C5H9NO2 |
| Molecular Weight | 115.133 |
| InChI Key | ONIBWKKTOPOVIA-UHFFFAOYSA-N |
| LogP | -1.64 |
| Synonyms |
|
Applications:
UV-Vis Spectrum of Proline
July 8, 2024
Access the UV-Vis Spectrum SIELC Library
If you are looking for optimized HPLC method to analyze Proline check our HPLC Applications library
For optimal results in HPLC analysis, it is recommended to measure absorbance at a wavelength that matches the absorption maximum of the compound(s) being analyzed. The UV spectrum shown can assist in selecting an appropriate wavelength for your analysis. Please note that certain mobile phases and buffers may block wavelengths below 230 nm, rendering absorbance measurement at these wavelengths ineffective. If detection below 230 nm is required, it is recommended to use acetonitrile and water as low UV-transparent mobile phases, with phosphoric acid and its salts, sulfuric acid, and TFA as buffers.

New HPLC Amino Acids Separation Compatible With Carbon Dating Technique
February 11, 2020
HPLC Method for Proline, Glycine, D-Alanine, Alanine, Hydroxyproline on Newcrom AH by SIELC Technologies
High Performance Liquid Chromatography (HPLC) Method for Analysis of Proline, Glycine, D-Alanine, Alanine, Hydroxyproline.
Hydroxyproline seems to be the most promising amino acid used in carbon dating when isolated from bone collagen. Separation of amino acids is challenging, especially without the use of ions or inorganic buffers that can interfere with Mass spectrometer (MS) or contaminate the sample with modern carbon. Amino acids are also not retained in reverse-phase chromatography. The ideal solution would be using water only to separate the amino acids. This would allow a direct coupling to MS. We were able to separate hydroxyproline from proline and other simple amino acids like glycine and alanine in HPLC on Newcrom AH column using water only as a mobile phase. Using water also allowed UV detection at 205 nm which can’t be done if using a buffer based on acetic or formic acid.
See more information on radiocarbon dating here.
The same method can be modified to get symmetrical peaks and higher efficiency if a mobile phase with ionic modifier such as formic acid is used.
You can find detailed UV spectra of Proline and information about its various lambda maxima by visiting the following link.
You can find detailed UV spectra of Glycine and information about its various lambda maxima by visiting the following link.
| Column | Newcrom AH, 4.6 x 150 mm, 5 µm, 100 A, dual ended |
| Mobile Phase | MeCN/H2O |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 205 nm, CAD |
| Class of Compounds |
Drug, Acid, Hydrophilic, Ionizable, Vitamin, Supplements, Amino acid |
| Analyzing Compounds | Proline, Glycine, D-Alanine, Alanine, Hydroxyproline |
Application Column
Newcrom AH
Column Diameter: 4.6 mm
Column Length: 150 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended
D-Alanine
Glycine
Hydroxyproline
Proline
CAD Detection

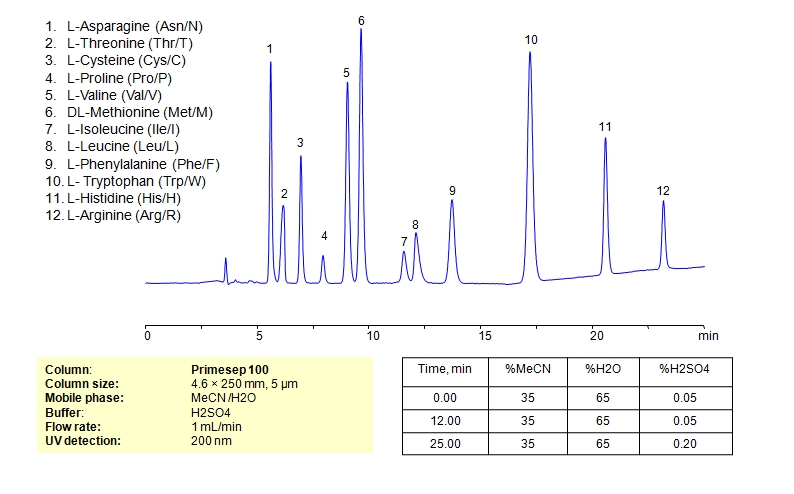
HPLC Separation of Mixture of 12 Amino Acids on Primesep 100 Column
March 11, 2019
HPLC Method for Asparagine, L-Cysteine, Cysteine, Proline, Valine, D-Valine, Methionine, L-Methionine, Isoleucine, D-Isoleucine, DL-Isoleucine, D-Leucine, Phenylalanine, Tryptophan, Histidine, Arginine, Amino Acids, Leucine, L-Threonine on Primesep 100 by SIELC Technologies
High Performance Liquid Chromatography (HPLC) Method for Analysis of Asparagine, L-Cysteine, Cysteine, Proline, Valine, D-Valine, Methionine, L-Methionine, Isoleucine, D-Isoleucine, DL-Isoleucine, D-Leucine, Phenylalanine, Tryptophan, Histidine, Arginine, Amino Acids, Leucine, L-Threonine.
Amino acids are the building blocks of proteins. Based on their dietary requirement, they are classified into essential and non-essential amino acids. Essential amino acids cannot be synthesized by the human body in sufficient quantities and must be obtained from the diet. Non-essential amino acids, on the other hand, can be synthesized by the body and are not dependent on dietary intake.
It’s worth noting that while these amino acids are considered “non-essential” for adults under normal circumstances because the body can synthesize them, there are situations where some may become “conditionally essential.” This means that under certain conditions like illness, stress, or trauma, the body might not produce them in sufficient quantities, and dietary intake becomes necessary. Arginine, for instance, is considered conditionally essential, especially during periods of rapid growth, illness, or trauma.
Amino acids can be retained, separeted and analyzed on a Primesep 100 mixed-mode stationary phase column using an isocratic analytical method with a simple mobile phase of water, Acetonitrile (MeCN), and a sulfuric acid (H2SO4) as a buffer. This analysis method can be detected in the UV regime at 200 nm.
| Column | Primesep 100, 4.6 x 250 mm, 5 µm, 100 A, dual ended |
| Mobile Phase | MeCN/H2O – 35/65% |
| Buffer | H2SO4 0.05% 12 min hold, gradient 0.05-0.20, 13 min |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 200 nm |
| Class of Compounds |
Drug, Acid, Hydrophilic, Ionizable, Vitamin, Supplements, Amino acid |
| Analyzing Compounds | Asparagine, L-Cysteine, Cysteine, Proline, Valine, D-Valine, Methionine, L-Methionine, Isoleucine, D-Isoleucine, DL-Isoleucine, D-Leucine, Phenylalanine, Tryptophan, Histidine, Arginine, Amino Acids, Leucine, L-Threonine |
Application Column
Primesep 100
Column Diameter: 4.6 mm
Column Length: 250 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended
Arginine
Asparagine
Cysteine
D-Isoleucine
D-Leucine
D-Valine
DL-Isoleucine
Histidine
Isoleucine
L-Cysteine
L-Methionine
L-Threonine
Leucine
Methionine
Phenylalanine
Proline
Tryptophan
Valine

HPLC Separation of Mixture of Conditionally Essential Amino Acids on Primesep 100 Column
March 11, 2019
HPLC Method for L-Glutamine, Glycine, Cysteine, L-Cysteine, Proline, Tyrosine, Arginine, Amino Acids on Primesep 100 by SIELC Technologies
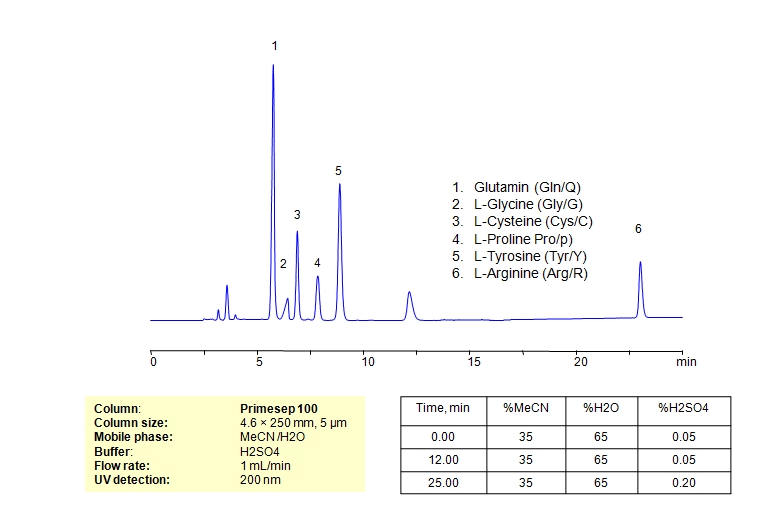
High Performance Liquid Chromatography (HPLC) Method for Analysis of L-Glutamine, Glycine, Cysteine, L-Cysteine, Proline, Tyrosine, Arginine, Amino Acids.
Glutamine is an amino acid with the chemical formula C5H10N2O3. It is a building block for protein, but it also supports the immune system, gut health, and detoxification. It is usually found in meat, fish, eggs, dairy, and whole grains; though some consider it the most abundant amino acid in the body. People also take glutamine as treatment for sickle cell disease, healing of burns, recovery after surgery, and injuries.
L-Glycine is an important amino acid compounds widely used in pharmaceutical, biochemical, and peptide research. It has he chemical formula C₂H₅NO₂. It is water-soluble and plays a critical role in protein synthesis, peptide modification, and metabolic studies. It can be found in meat., eggs. and bones.
L-Cysteine is an amino acid with the chemical formula C5H10N2O3. It is primarily a building block for protein, but it also has antioxidant effects. On occasion, ot os used to support people dealing with cancer, diabetes, and hangover, but there is yet to be substantial evidence that it works. Poultry, egg, beef, and whole grains are rich sources of the amino acid.
L-Proline is an amino acid with the chemical formula C5H9NO2. It is naturally produced in the body, but it is also recommended to be consumed through the diet. It can be found in meat, fish, and dairy. It is used for skin healing and treatment of other skin conditions as it is an essential component of collagen. It is important for upkeep of joints and tendons, as well as heart muscles.
L-Tyrosine is an amino acid with the chemical formula C9H11NO3. It is naturally used in the body to create neurotransmitters like dopamine and norepinephrine. While it is produced in the body naturally, it can also be consumed in beef, pork, fish, dairy products, and beans. As a supplement, it is also used to treat Phenylketonuria (PKU).
L-Arginine is an amino acid with the chemical formula C9H11NO3. It is a precursor for nitric acid, making it crucial for blood flow and general cardiovascular health. It can help lower blood pressure, help with chest pain, and improve symptoms of Peripheral arterial disease (PAD).
L-Glutamine, Glycine, Cysteine, L-Cysteine, Proline, Tyrosine, Arginine, Amino Acids can be retained and analyzed using the Primesep 100 stationary phase column. The analysis utilizes an isocratic method with a simple mobile phase consisting of water and acetonitrile (MeCN) with a sulfuric acid buffer. Detection is performed using UV.
| Column | Primesep 100, 4.6 x 250 mm, 5 µm, 100 A, dual ended |
| Mobile Phase | MeCN/H2O – 35/65% |
| Buffer | H2SO4 0.05% 12 min hold, gradient 0.05-0.20, 13 min |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 200 nm |
| Class of Compounds |
Drug, Acid, Hydrophilic, Ionizable, Vitamin, Supplements, Amino acid |
| Analyzing Compounds | L-Glutamine, Glycine, Cysteine, L-Cysteine, Proline, Tyrosine, Arginine, Amino Acids |
Application Column
Primesep 100
Column Diameter: 4.6 mm
Column Length: 250 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended
Arginine
Cysteine
Glycine
L-Cysteine
L-Glutamine
Proline
Tyrosine

HPLC Separation of Lysine and Arginine from Other Amino Acids
July 10, 2012
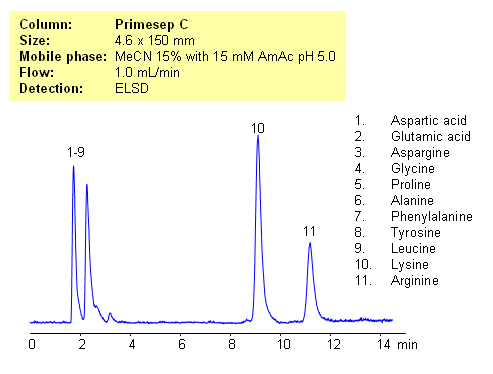
Application Notes: Amino acids are polar ionic compounds which are not retained on reversed-phase column without ion-pairing reagent. In our application, lysine and arginine can be separated from other amino acids. Amino acids with a pH between 3 and 5 and with one basic and one acidic group become very polar. Therefore these amino acids don’t have strong ion-exchange interaction with Primesep C stationary phase. Amino acids with two amino groups still carry positive net charge and can interact with stationary phase by cations-exchange mechanism. pH variation of the mobile phase can be an effective tool to adjust selectivity of separation for zwitter-ionic, basic and acidic compounds. This method can be used for separation of mono-charged compounds from compounds having an extra charge.
Application Columns: Primesep C
Application compounds: Aspartic acid, Glutamic acid, Aspargine, Glycine, Proline, Alanine, Phenylalanine, Tyrosine, Leucine, Lysine, Arginine
Detection technique: UV, LC/MS, ELSD/CAD
| Column | Primesep C, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN – 15% |
| Buffer | AmAc pH 5.0- 15 mM |
| Flow Rate | 1.0 ml/min |
| Detection | ELSD |
| Class of Compounds |
Drug, Acid, Hydrophilic, Ionizable, Vitamin, Supplements |
| Analyzing Compounds | Aspartic acid, Glutamic acid, Aspargine, Glycine, Proline, Alanine, Phenylalanine, Tyrosine, Leucine, Lysine, Arginine |
Application Column
Primesep C
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsArginine
Asparagine
Aspartic Acid
Glutamic Acid
Glycine
Leucine
Lysine
Phenylalanine
Proline
Tyrosine
UV Detection

HPLC Analysis of Active Drug and Amino Acids in a Formulation
October 14, 2010
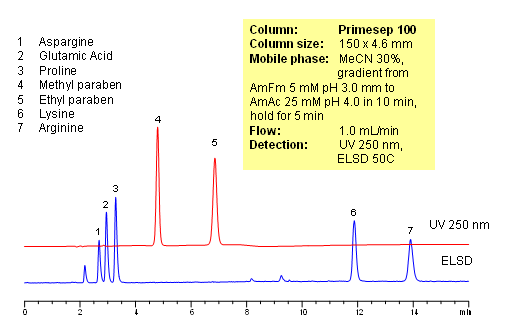
Polar amino acids are very often used as components of vitamin and supplement composition. Analysis of such complex composition is a challenging task. In this application, 5 amino acids (asparagine, glutamic acid, proline and arginine) and two preservatives (methyl paraben and propyl paraben) are separated on a Primesep 100 reversed-phase cation-exchange column with LC/MS compatible mobile phase. Method does not require ion-pairing reagent in the mobile phase. Compounds are monitored by ELSD and UV. Method is validated for quantitation of underivatized amino acids in complex mixtures. The method is simple and robust and can be used for analysis of various vitamin formulations.
| Column | Primesep 100, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | AmFm |
| Flow Rate | 1.0 ml/min |
| Detection | ELSD 50C, UV 250 nm |
| Class of Compounds |
Drug, Acid, Hydrophilic, Ionizable, Vitamin, Supplements |
| Analyzing Compounds | Glutamic acid, Aspargine, Proline, Lysine, Arginine, Methyl paraben, Propyl paraben |
Application Column
Primesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsAsparagine
Ethylparaben
Glutamic Acid
Lysine
Methylparaben
Proline
UV Detection

HPLC Analysis of Cheese Extract using Mixed-Mode Chromatography
May 6, 2005
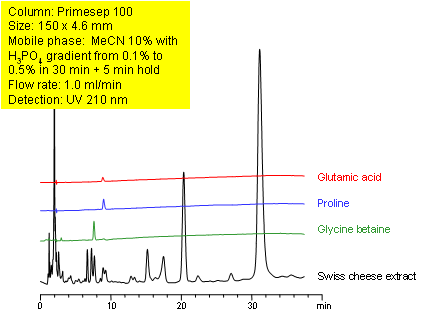
Primesep 100 separates the components of a Swiss cheese extract by HPLC using cation exchange and reversed phase as retention mechanisms. The amino acids, glutamic acid and proline, as well as glycine betaine were resolved in less than 10 minutes. A mobile phase gradient of water, acetonitrile (MeCN, ACN,) and phosphoric acid (H3PO4) and ultraviolet (UV) detection was used.
| Column | Primesep 100, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | H3PO4 |
| Flow Rate | 1.0 ml/min |
| Detection | UV 210 nm |
| Class of Compounds |
Drug, Acid, Hydrophilic, Ionizable, Vitamin, Supplements |
| Analyzing Compounds | Glutamic acid, Proline, Glycine betaine |
Application Column
Primesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsGlutamic Acid
Glycine Betaine
Proline