| CAS Number | 63-68-3 |
|---|---|
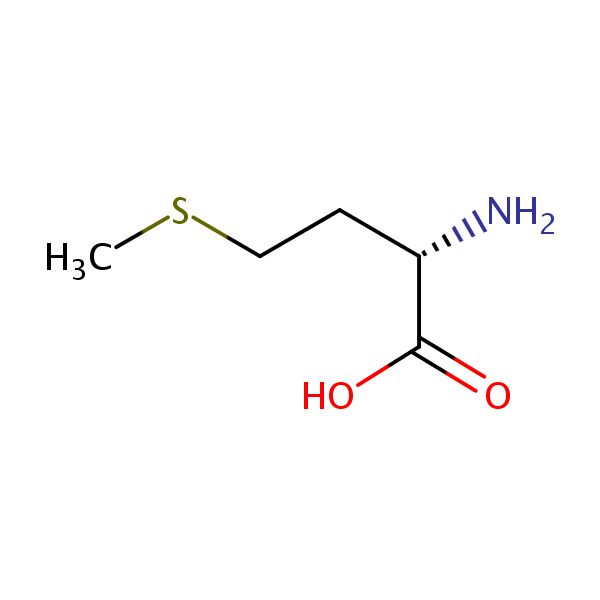
| Molecular Formula | C5H11NO2S |
| Molecular Weight | 149.211 |
| InChI Key | FFEARJCKVFRZRR-BYPYZUCNSA-N |
| LogP | -1.87 |
| Synonyms |
|
Applications:
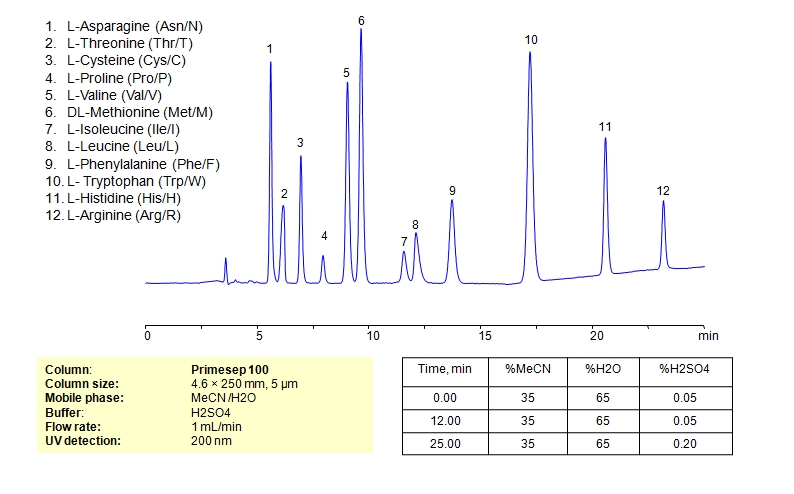
HPLC Separation of Mixture of 12 Amino Acids on Primesep 100 Column
March 11, 2019
| Column | Primesep 100, 4.6×250 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O – 35/65% |
| Buffer | H2SO4 0.05% 12 min hold, gradient 0.05-0.20, 13 min |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 200 nm |
| Class of Compounds |
Drug, Acid, Hydrophilic, Ionizable, Vitamin, Supplements, Amino acid |
| Analyzing Compounds | L-Asparagine (Asn/N), L-Threonine (Thr/T), L-Cysteine (Cys/C), L-Proline (Pro/P), L-Valine (Val/V), DL-Methionine (Met/M), L-Isoleucine (Ile/I), L-Leucine (Leu/L), L-Phenylalanine (Phe/F), L- Tryptophan (Trp/W), L-Histidine (His/H), L-Arginine (Arg/R) |
Application Column
Primesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsArginine
Asparagine
Cysteine
D-Isoleucine
D-Leucine
D-Valine
DL-Isoleucine
Histidine
Isoleucine
L-Cysteine
L-Methionine
L-Threonine
Leucine
Methionine
Phenylalanine
Proline
Tryptophan
Valine

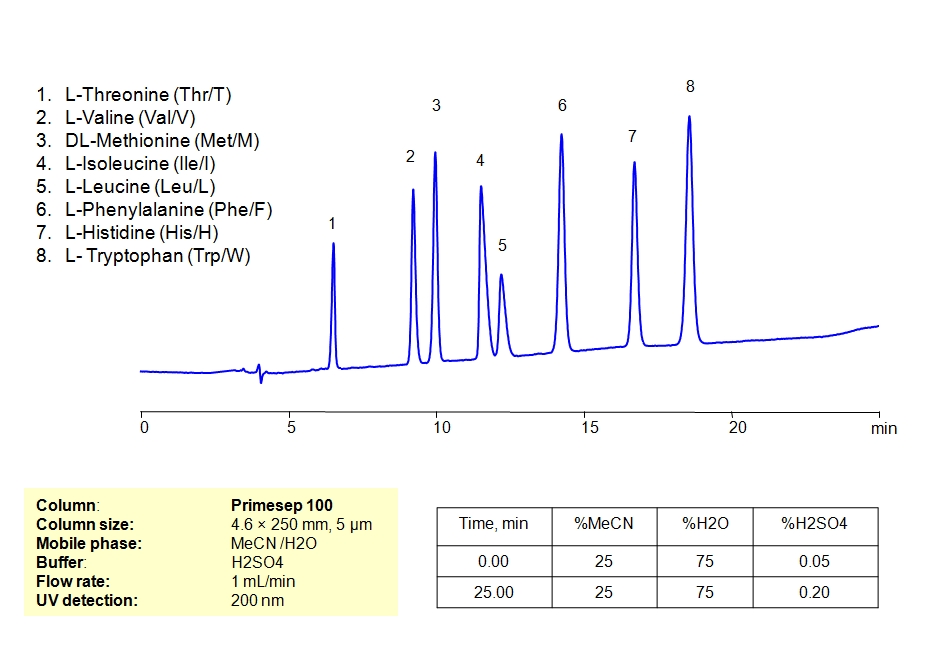
HPLC Separation of Mixture of Essential Amino Acids on Primesep 100 Column
March 11, 2019
| Column | Primesep 100, 4.6×250 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O – 25/75% |
| Buffer | Gradient H2SO4 0.05-0.2% 25 min |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 200 nm |
| Class of Compounds |
Drug, Acid, Hydrophilic, Ionizable, Vitamin, Supplements, Amino acid |
| Analyzing Compounds | L-Threonine (Thr/T), L-Valine (Val/V), DL-Methionine (Met/M), L-Isoleucine (Ile/I), L-Leucine (Leu/L), L-Phenylalanine (Phe/F), L-Histidine (His/H), L- Tryptophan (Trp/W) |
Application Column
Primesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsD-Isoleucine
D-Leucine
D-Valine
DL-Isoleucine
Histidine
Isoleucine
L-Histidine hydrochloride monohydrate
L-Isoleucine
L-Methionine
L-Threonine
Methionine
Phenylalanine
Tryptophan
Valine

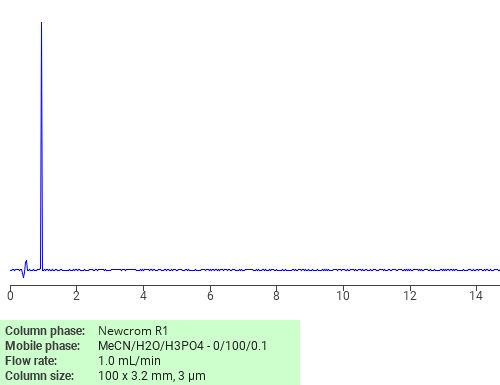
Separation of L-Methionine on Newcrom R1 HPLC column
February 16, 2018
L-Methionine can be analyzed by this reverse phase (RP) HPLC method with simple conditions. The mobile phase contains an acetonitrile (MeCN), water, and phosphoric acid. For Mass-Spec (MS) compatible applications the phosphoric acid needs to be replaced with formic acid. Smaller 3 µm particles columns available for fast UPLC applications. This liquid chromatography method is scalable and can be used for isolation impurities in preparative separation. It also suitable for pharmacokinetics.
Application Column
Newcrom R1
The Newcrom columns are a family of reverse-phase-based columns. Newcrom A, AH, B, and BH are all mixed-mode columns with either positive or negative ion-pairing groups attached to either short (25 Å) or long (100 Å) ligand chains. Newcrom R1 is a special reverse-phase column with low silanol activity.
Select options