| CAS Number | 4350-09-8 |
|---|---|
| Molecular Formula | C11H12N2O3 |
| Molecular Weight | 220.228 |
| InChI Key | LDCYZAJDBXYCGN-VIFPVBQESA-N |
| LogP | -0.735 |
| Synonyms |
|
Applications:
Generic Screening Method for Complex Mixtures on Primesep 200
October 15, 2015
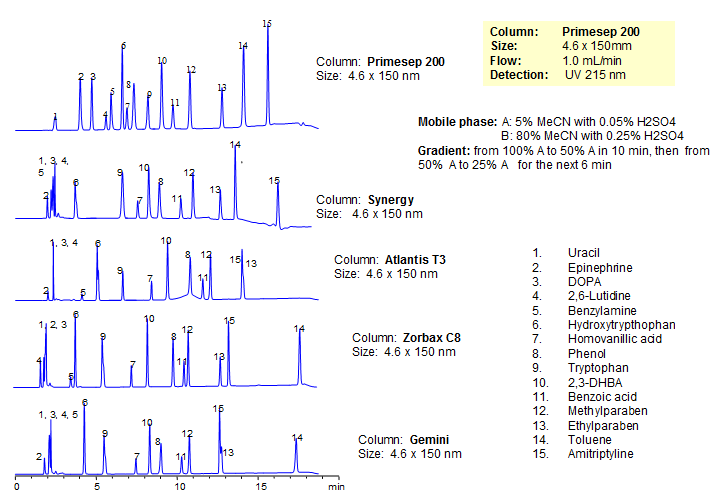
| Column | Primesep 200, 4.6*150 mm 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | H2SO4 |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 215 nm |
| Class of Compounds |
Drug, Acid, Hydrophilic, Ionizable, Hormone |
| Analyzing Compounds | Uracil, Epinephrine, DOPA, 2,6-Lutidine, Benzylamine, Hydroxytrypthophan, Homovanillic acid, Phenol, Tryptophan , 2,3-DHBA, Benzoic acid, Methylparaben, Ethylparaben, Toluene, Amitriptyline |
Application Column
Primesep 200
Column Diameter: 4.6 mm
Column Length: 150 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended
2,6-Lutidine
Amitriptyline
Benzoic Acid
Benzylamine
DOPA (3,4-dihydroxy-L-phenylalanine)
Epinephrine
Ethylparaben
Homovanillic Acid
Hydroxytryptophan
Methylparaben
Phenol
Toluene
Tryptophan
Uracil

HPLC Separation of Neurotransmitters
November 25, 2011
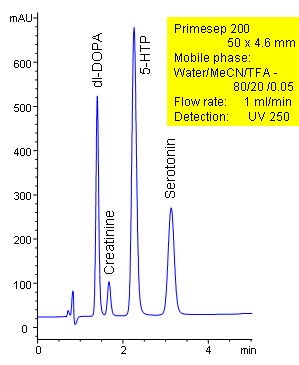
The neurotransmitters dl-DOPA, creatinine, hydroxytryptophan (5-HTP), and serotonin are separated in less than 4 minutes on a short 50 mm Primesep 200 column. The HPLC separation uses a mobile phase of water, acetonitrile (MeCN, ACN) and trifluoroacetic acid (TFA) and UV detection at 250 nm. Ion-pair reagents were not needed for retention of these polar, hydrophilic compounds, instead, a combination of ion-exchange and reversed-phase interactions were used.
| Column | Primesep 200, 4.6×50 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O – 80/20% |
| Buffer | TFA – 0.05% |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 250 nm |
| Class of Compounds |
Drug, Acid, Hydrophilic, Ionizable, Hormone |
| Analyzing Compounds | dl-DOPA, Creatinine, Hydroxytryptophan (5-HTP), Serotonin |
Application Column
Primesep 200
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsDOPA (3,4-dihydroxy-L-phenylalanine)
Hydroxytryptophan
Serotonin

Separation of Serotonin, Dopamine, and Related Compounds
August 22, 2008
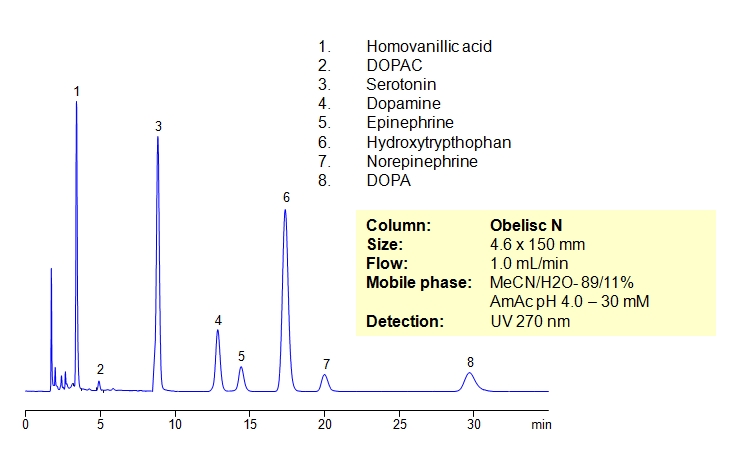
Catecholamines are chemical compounds derived from the amino acid tyrosine containing catechol and amine groups. Some of them are biogenic amines. Retention of compounds of the catecholamine pathway is achieved on Obelisc N column. All polar compounds are well retained by combination of HILIC and ion-exchange mechanisms. Obelisc N columns produce very good peak shapes for all analytes. The method is very sensitive to amount of ACN, buffer and buffer pH. The retention time changes with variation of the main parameters. This method can be used for quantitation of biogenic amines and related compounds (homovanillic acid, dihydroxyphenyl acetic acid, serotonin, dopamine, epinephrine, hydroxytryptophan, epinephrine and DOPA) in urine, blood and other biological fluids. Further optimization of this HPLC method can be used during screening and validation. Amines and acids can be analyzed in the same run and retained by a combination of polar organic mode, cation-exchange and anion-exchange modes. Various buffers within specified pH can be employed (ammonium formate, ammonium acetate, sodium phosphate, etc.).
| Column | Obelisc N, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | AmAc pH 4.0- 30 mM |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 270 nm |
| Class of Compounds |
Drug, Acid, Monocarboxylic acid, Hydrophilic, Ionizable, Hormone |
| Analyzing Compounds | Homovanillic acid, Dihydroxyphenyl acetic acid, Serotonin, Dopamine, Epinephrine, Hydroxytryptophan, DOPA |
Application Column
Obelisc N
SIELC has developed the Obelisc™ columns, which are mixed-mode and utilize Liquid Separation Cell technology (LiSC™). These cost-effective columns are the first of their kind to be commercially available and can replace multiple HPLC columns, including reversed-phase (RP), AQ-type reversed-phase, polar-embedded group RP columns, normal-phase, cation-exchange, anion-exchange, ion-exclusion, and HILIC (Hydrophilic Interaction Liquid Chromatography) columns. By controlling just three orthogonal method parameters - buffer concentration, buffer pH, and organic modifier concentration - users can adjust the column properties with pinpoint precision to separate complex mixtures.
Select optionsDOPAC (Dihydroxyphenylacetic Acid)
Dopamine
Epinephrine
Homovanillic Acid
Hydroxytryptophan
Norepinephrine
Serotonin