| CAS Number | 90-64-2 |
|---|---|
| Molecular Formula | C8H8O3 |
| Molecular Weight | 152.149 |
| InChI Key | IWYDHOAUDWTVEP-UHFFFAOYSA-N |
| LogP | 0.62 |
| Synonyms |
|
Applications:
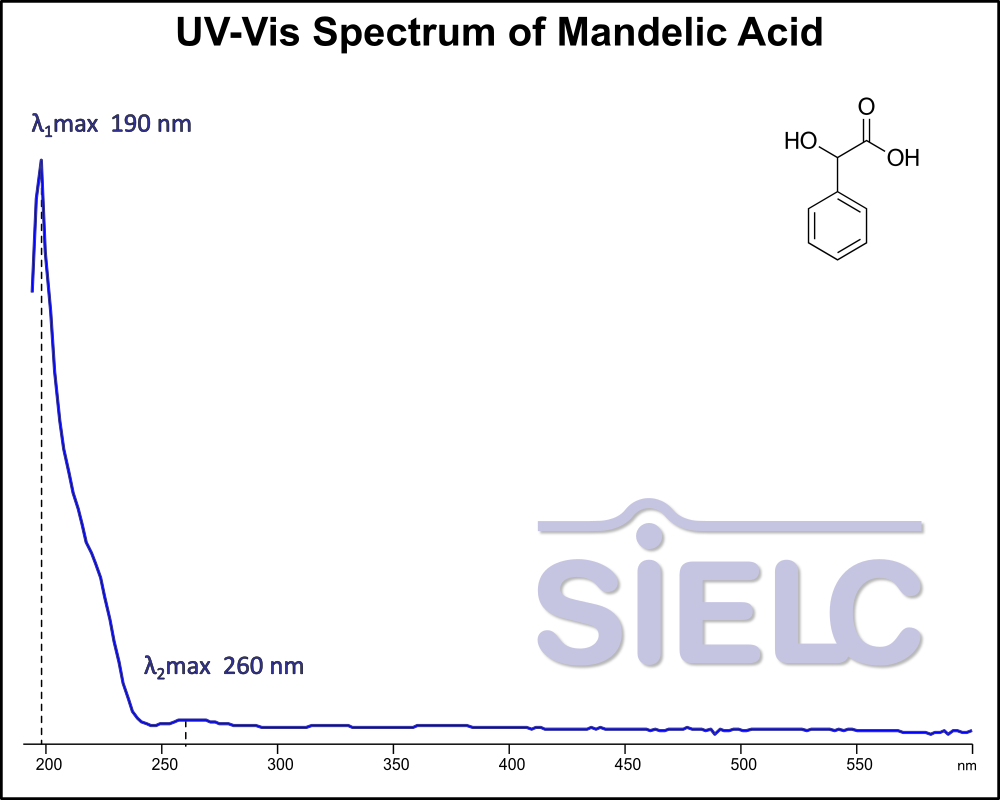
UV-Vis Spectrum of Mandelic Acid
December 18, 2025
If you are looking for optimized HPLC method to analyze Mandelic Acid check our HPLC Applications library
For optimal results in HPLC analysis, it is recommended to measure absorbance at a wavelength that matches the absorption maximum of the compound(s) being analyzed. The UV spectrum shown can assist in selecting an appropriate wavelength for your analysis. Please note that certain mobile phases and buffers may block wavelengths below 230 nm, rendering absorbance measurement at these wavelengths ineffective. If detection below 230 nm is required, it is recommended to use acetonitrile and water as low UV-transparent mobile phases, with phosphoric acid and its salts, sulfuric acid, and TFA as buffers.
For some compounds, the UV-Vis Spectrum is affected by the pH of the mobile phase. The spectra presented here are measured with an acidic mobile phase that has a pH of 3 or lower.

HILIC Separation of Carboxylic Acids
August 22, 2008
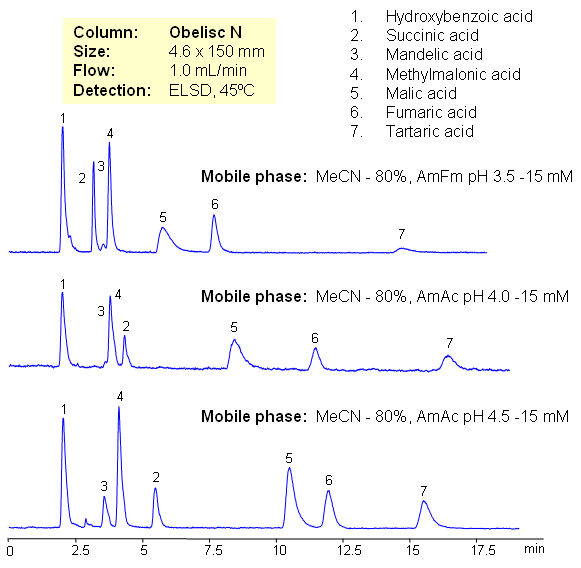
Hydrophilic acids are separated on Obelisc N mixed-mode HILIC column. Seven carboxylic acids are separated based on their polarity and pKa values. Changes in ionization states of acids and stationary phase can be used to control elution order of organic and inorganic acids.
Application Column
Obelisc N
SIELC has developed the Obelisc™ columns, which are mixed-mode and utilize Liquid Separation Cell technology (LiSC™). These cost-effective columns are the first of their kind to be commercially available and can replace multiple HPLC columns, including reversed-phase (RP), AQ-type reversed-phase, polar-embedded group RP columns, normal-phase, cation-exchange, anion-exchange, ion-exclusion, and HILIC (Hydrophilic Interaction Liquid Chromatography) columns. By controlling just three orthogonal method parameters - buffer concentration, buffer pH, and organic modifier concentration - users can adjust the column properties with pinpoint precision to separate complex mixtures.
Select optionsHydroxybenzoic Acid
Malic Acid
Mandelic Acid
Methylmalonic Acid
Organic Acids
Succinic Acid
Tartaric Acid

Complex Mixture of Acids, Bases, Amino Acids, and Neutral Compounds
October 14, 2006
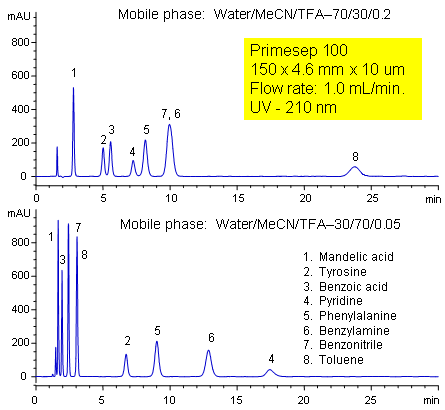
Primesep 100 separates a mixture of amino acids (tyrosine, phenylalanine), organic acids (benzoic acid, mandelic acid), amines (benzylamine, pyridine), and neutrals (benzonitrile, toluene) in one HPLC run by combining reversed-phase, cation-exchange, and polar interactions. The method is tunable and peak order can be changed significantly by adjusting acetonitrile and trifluoroacetic acid concentrations. The separation method uses a mobile phase mixture of water, acetonitrile (MeCN, ACN) and trifluoracetic acid (TFA) and compatible with UV, mass spec (LC/MS) and evaporative light scattering (ELSD) detection.
| Column | Primesep 100, 4.6×250 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O – 30/70% |
| Buffer | TFA – 0.2 |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 210 nm |
| Class of Compounds |
Drug, Acid, Hydrophilic, Ionizable, Vitamin, Supplements, Amino acid |
| Analyzing Compounds | Tyrosine, phenylalanine, Benzoic acid, mandelic acid, Benzylamine, Pyridine, Benzonitrile, Toluene |
Application Column
Primesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsBenzoic Acid
Benzonitrile
Benzylamine
Mandelic Acid
Organic Acids
Phenylalanine
Pyridine
Toluene
Tyrosine