| CAS Number | 69-72-7 |
|---|---|
| Molecular Formula | C7H6O3 |
| Molecular Weight | 138.122 |
| InChI Key | YGSDEFSMJLZEOE-UHFFFAOYSA-N |
| LogP | 2.26 |
| Synonyms |
|
Applications:
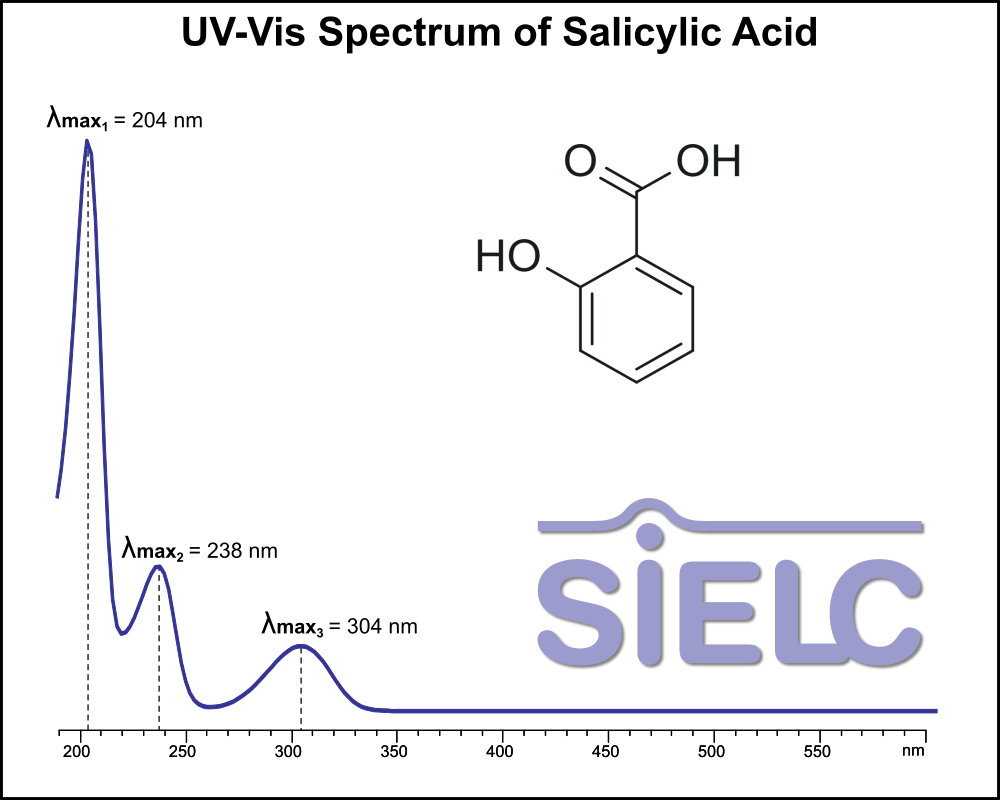
UV-Vis Spectrum of Salicylic Acid
July 31, 2025
If you are looking for optimized HPLC method to analyze Salicylic acid check our HPLC Applications library
For optimal results in HPLC analysis, it is recommended to measure absorbance at a wavelength that matches the absorption maximum of the compound(s) being analyzed. The UV spectrum shown can assist in selecting an appropriate wavelength for your analysis. Please note that certain mobile phases and buffers may block wavelengths below 230 nm, rendering absorbance measurement at these wavelengths ineffective. If detection below 230 nm is required, it is recommended to use acetonitrile and water as low UV-transparent mobile phases, with phosphoric acid and its salts, sulfuric acid, and TFA as buffers.
For some compounds, the UV-Vis Spectrum is affected by the pH of the mobile phase. The spectra presented here are measured with an acidic mobile phase that has a pH of 3 or lower.

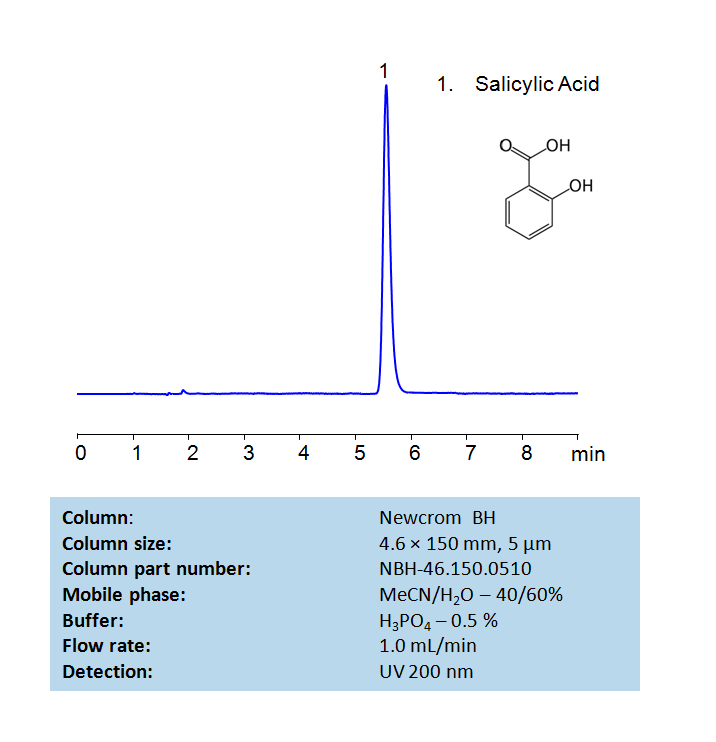
HPLC Method For Analysis Of Salicylic Acid on Newcrom BH Column
May 2, 2022
Separation type: Liquid Chromatography Mixed-mode
Salicylic acid (SA) is an o-hydroxybenzoic acid, synthesized by both plants and microorganisms. SA acts as a critical plant hormone regulating various processes, including growth and development, flowering, thermogenesis, ion uptake, stomatal movement, photosynthesis, and plant immunity. SA also has medical applications as a topical treatment for a wide variety of skin conditions, including warts, dandruff, and acne. Using a Newcrom BH mixed-mode column and a mobile phase consisting of water, Acetonitrile (MeCN), and a Phosphoric acid (H3PO4) buffer, Salicylic acid can be retained, measured, and analyzed. This analysis method can be UV detected at 200 nm with high resolution with high resolution and peak symmetry.
High Performance Liquid Chromatography (HPLC) Method for Analysis of Salicylyc Acid
| Column | Newcrom BH, 4.6×150 mm, 100A |
| Mobile Phase | MeCN – 40% |
| Buffer | H3PO4 – 0.5% |
| Flow Rate | 1.0 ml/min |
| Detection | UV 200nm |
| Class of Compounds | Acid, Hydrophilic |
| Analyzing Compounds | Salicylic Acid |
Application Column
Newcrom BH
The Newcrom columns are a family of reverse-phase-based columns. Newcrom A, AH, B, and BH are all mixed-mode columns with either positive or negative ion-pairing groups attached to either short (25 Å) or long (100 Å) ligand chains. Newcrom R1 is a special reverse-phase column with low silanol activity.
Select options
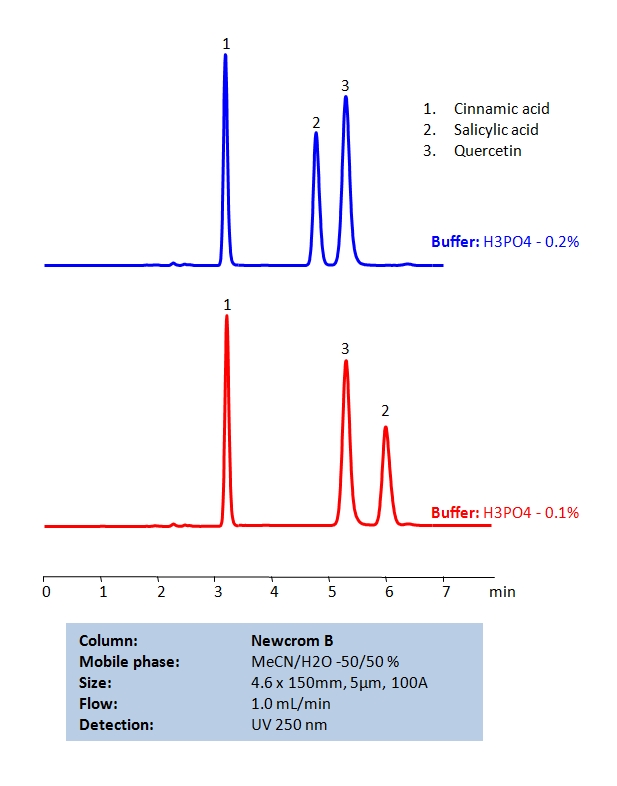
HPLC Separation of Cinnamic Acid, Salicylic Acid, and Quercetin on Newcrom B Column
March 12, 2021
Method for Cinnamic acid, Salicylic acid, Quercetin, on Newcrom B by SIELC Technologies
High Performance Liquid Chromatography (HPLC) Method of Cinnamic Acid, Salicylic Acid and Quercetin.
Cinnamic acid and benzoic acid are two of the major phenolic acids and autotoxins. Cinnamic acid occurs naturally in all green plants, showing significant antimicrobial activity against both bacteria and fungi as well as antioxidant activity. It has the chemical formula C9H8O2.
Salicylic acid (SA) is an o-hydroxybenzoic acid, synthesized by both plants and microorganisms. SA acts as a critical plant hormone regulating various processes, including growth and development, flowering, thermogenesis, ion uptake, stomatal movement, photosynthesis, and plant immunity. It has the chemical formula C7H6O3.
Quercetin is a naturally occurring flavonol, or flavonoid, the yellowish antioxidant pigment found in skins of red grapes, berries, apples, onions, tomatoes, and buckwheat tea. In addition to functioning as a flavonoid, quercetin is also a phytoestrogen. It has the chemical formula C15H10O7.
Cinnamic acid, Salicylic acid, Quercetin, can be retained and separated by using a mixed-mode Newcrom B column using an isocratic analytical method with a simple mobile phase of water, acetonitrile (MeCN, ACN), and phosphoric acid (H3PO4) buffer. UV detection at 250 nm.
| Column | Newcrom B, 4.6 x 150 mm, 5 µm, 100 A, dual ended |
| Mobile Phase | MeCN -50% |
| Buffer | H3PO4 |
| Flow Rate | 1.0 ml/min |
| Detection | 250 nm |
| Class of Compounds | Acid |
| Analyzing Compounds | Cinnamic acid, Salicylic acid, Quercetin, |
Application Column
Newcrom B
Column Diameter: 4.6 mm
Column Length: 150 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended
Cinnamic acid
Quercetin
Salicylic acid

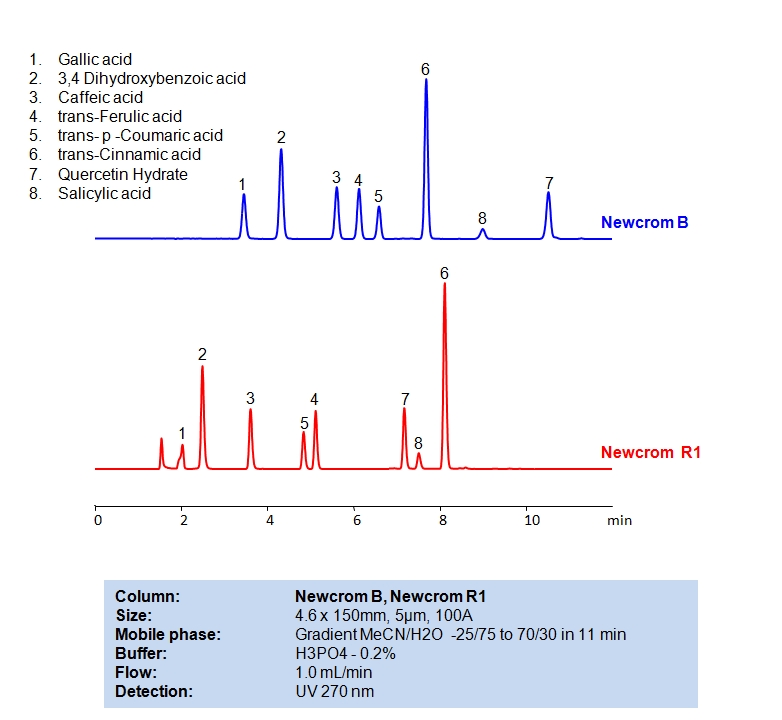
HPLC Separation of Phenolic Acids
March 11, 2021
HPLC Method for Gallic acid, 3,4-Dihydroxybenzoic Acid, Caffeic acid, Ferulic acid, p-coumaric acid, Cinnamic acid, Salicylic acid, Quercetin on Newcrom B by SIELC Technologies
High Performance Liquid Chromatography (HPLC) Method for Analysis of Gallic acid, 3,4-Dihydroxybenzoic Acid, Caffeic acid, Ferulic acid, p-coumaric acid, Cinnamic acid, Salicylic acid, Quercetin.
Gallic acid is a naturally-occurring phenolic acid that was a key ingredient in European iron gall ink from its first discovery in the 12th century until the 19th century. In modern times, it’s heavy metal salt, Bismuth subgallate, known by its trade name Devrom, is known for its effects at deodorizing flatulence and stools. It has the chemical formula C7H6O5.
3,4 Dihydroxybenzoic acid, also known as Protocatechuic acid (PCA), is a dihydroxybenzoic acid with the chemical formula C7H6O4. It occurs naturally in stem bark of Boswellia dalzielii, leaves of Diospyros melanoxylon, Açai oil, and roselle. Commercially , it is produced of vanillin. You can find detailed UV spectra of 3,4 Dihydroxybenzoic acid and information about its various lambda maxima by visiting the following link.
Caffeic acid is an organic compound with the formula (HO)2C6H3CH=CHCO2H. It plays a key role in scavenging reactive oxygen species (ROS) that are generated during energy metabolism. Caffeic acid is an active ingredient in caffenol, which is a photographic developer made from instant coffee. Pharmacologically, it is used as an antioxidant in vitro and also in vivo. It also shows immunomodulatory and anti-inflammatory activity.
trans-Ferulic acid is a hydroxycinnamic acid derivative with the chemical formula C10H10O4. It is classified as a phenolic phytochemical. Naturally, it can be found in a large number of vegetables, grain, and other plants. It is also released while sweetcorn is being cooked. Salts and esters derived from ferulic acid are called ferulates.
trans-p-Coumaric acid is a major phenolic acid with the chemical formula C9H8O3. It can be found in a wide variety of graminaceous plants. p-Coumaric acid is a natural metabolite contained in many edible plants and its antioxidant activities in reducing oxidative stress and inflammatory reactions have been demonstrated in various experiments.
trans-Cinnamic acid and benzoic acid are two of the major phenolic acids and autotoxins. Cinnamic acid occurs naturally in all green plants, showing significant antimicrobial activity against both bacteria and fungi as well as antioxidant activity. It has the chemical formula C9H8O2.
Quercetin Hydrate is a naturally occurring flavonol, or flavonoid, the yellowish antioxidant pigment found in skins of red grapes, apples, berries, onions, tomatoes, and buckwheat tea. In addition to functioning as a flavonoid, quercetin is also a phytoestrogen. It has the chemical formula C15H10O7.
Salicylic acid (SA) is an o-hydroxybenzoic acid, synthesized by both plants and microorganisms. SA acts as a critical plant hormone regulating various processes, including growth and development, flowering, thermogenesis, ion uptake, stomatal movement, photosynthesis, and plant immunity. SA also has medical applications as a topical treatment for a wide variety of skin conditions, including warts, dandruff, and acne.
Gallic acid, 3,4-Dihydroxybenzoic Acid, Caffeic acid, Ferulic acid, p-coumaric acid, Cinnamic acid, Salicylic acid, Quercetin can be retained and analyzed using the Newcrom B stationary phase column. The analysis utilizes an isocratic method with a simple mobile phase consisting of water and acetonitrile (MeCN) with a phosphoric acid buffer. Detection is performed using UV.
| Column | Newcrom B, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | Gradient MeCN |
| Buffer | H3PO4 – 0.2% |
| Flow Rate | 1.0 ml/min |
| Detection | 270 nm |
| Column | Newcrom R1, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | Gradient MeCN |
| Buffer | H3PO4 – 0.2% |
| Flow Rate | 1.0 ml/min |
| Detection | 270 nm |
| Class of Compounds | Acid |
| Analyzing Compounds | Gallic acid, 3,4-Dihydroxybenzoic Acid, Caffeic acid, Ferulic acid, p-coumaric acid, Cinnamic acid, Salicylic acid, Quercetin |
Application Column
Newcrom B
Column Diameter: 4.6 mm
Column Length: 150 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended
Newcrom R1
Column Diameter: 4.6 mm
Column Length: 150 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended
Caffeic acid
Cinnamic acid
Ferulic acid
Gallic acid
Quercetin
Salicylic acid
p-coumaric acid

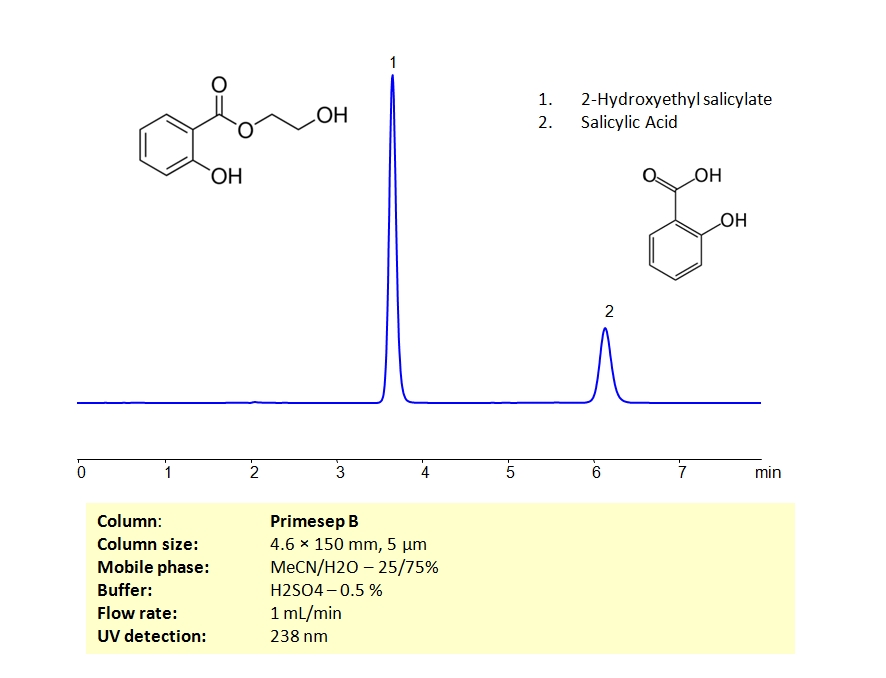
HPLC Separation of Mixture of 2-Hydroxyethyl Salicylate and Salicylic Acid on Primesep B Column
August 14, 2019
HPLC Method for 2-Hydroxyethyl salicylate, Salicylic acid on Primesep B by SIELC Technologies
High Performance Liquid Chromatography (HPLC) Method for Analysis of 2-Hydroxyethyl salicylate, Salicylic acid
2-Hydroxyethyl salicylate is a drug used as analgesic agents for the treatment of mild to moderate pain.
2-Hydroxyethyl salicylate, Salicylic acid can be retained and analyzed using the Primesep B stationary phase column. The analysis utilizes an isocratic method with a simple mobile phase consisting of water and acetonitrile (MeCN) with a sulfuric acid buffer. Detection is performed using UV.
| Column | Primesep B |
| Mobile Phase | MeCN/H2O – 25/75% |
| Buffer | H2SO4 – 0.5 % |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 238 nm |
| Class of Compounds |
Acid, Hydrophilic, Ionizable, Carboxylic acid, Drug, Carbocyclic, Benzoate, Hydroxy Acid, Hydroxybenzoate, Phenol |
| Analyzing Compounds | 2-Hydroxyethyl salicylate, Salicylic acid |
Application Column
Primesep B
Column Diameter: 4.6 mm
Column Length: 150 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended
Salicylic acid

HPLC Separation of Salicylic acid
August 6, 2015
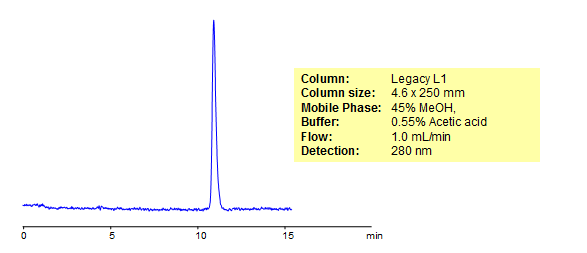
Salicylic acid is a pain-reliever and anti-inflammatory commonly found in rubefacient products for topical pain relief as well as cosmetic skin-care products used to treat a variety of skin conditions. Legacy L1 was used to retain salicylic acid by reverse phase mechanism.Legacy L1 uses embedded C18 groups on porous silica and is useful for many USP HPLC applications. comparisons to Phenomenex columns are available by request.
| Column | Legacy L1, 4.6×250 mm, 5 µm, 100A |
| Mobile Phase | MeOH – 45% |
| Buffer | Acetic acid – 0.55% |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 280 nm |
| Class of Compounds |
Acid, Hydrophilic, Ionizable |
| Analyzing Compounds | Salicylic acid |
Application Column
Legacy L1
SIELC's family of Legacy columns is based on the United States Pharmacopeia's (USP) published chromatographic methods and procedures. Numerous brands have columns used in USP reference standards and methods. USP has created various designations to group together columns with similar types of packing and properties in the solid phase. SIELC's Legacy columns adhere to these strict requirements and properties, allowing you to easily replace older columns that are no longer available without needing to significantly modify your method or SOPs.
Select options