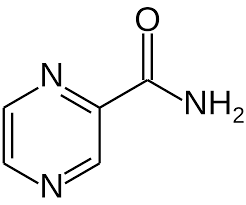
| CAS Number | 98-96-4 |
|---|---|
| Molecular Formula | C5H5N3O |
| Molecular Weight | 123.11 |
| InChI Key | IPEHBUMCGVEMRF-UHFFFAOYSA-N |
| LogP | -0.6 |
| Synonyms |
|
Applications:
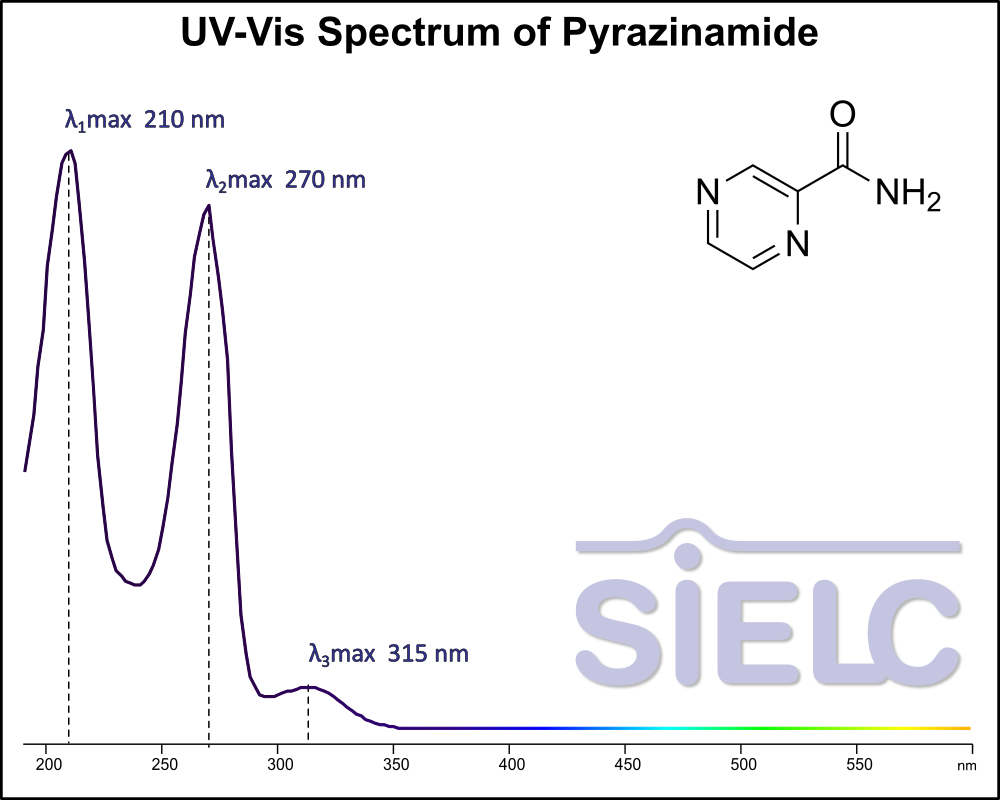
Uv-Vis Spectrum of Pyrazinamide
January 19, 2026
If you are looking for optimized HPLC method to analyze Pyrazinamide check our HPLC Applications library
For optimal results in HPLC analysis, it is recommended to measure absorbance at a wavelength that matches the absorption maximum of the compound(s) being analyzed. The UV spectrum shown can assist in selecting an appropriate wavelength for your analysis. Please note that certain mobile phases and buffers may block wavelengths below 230 nm, rendering absorbance measurement at these wavelengths ineffective. If detection below 230 nm is required, it is recommended to use acetonitrile and water as low UV-transparent mobile phases, with phosphoric acid and its salts, sulfuric acid, and TFA as buffers.
For some compounds, the UV-Vis Spectrum is affected by the pH of the mobile phase. The spectra presented here are measured with an acidic mobile phase that has a pH of 3 or lower.

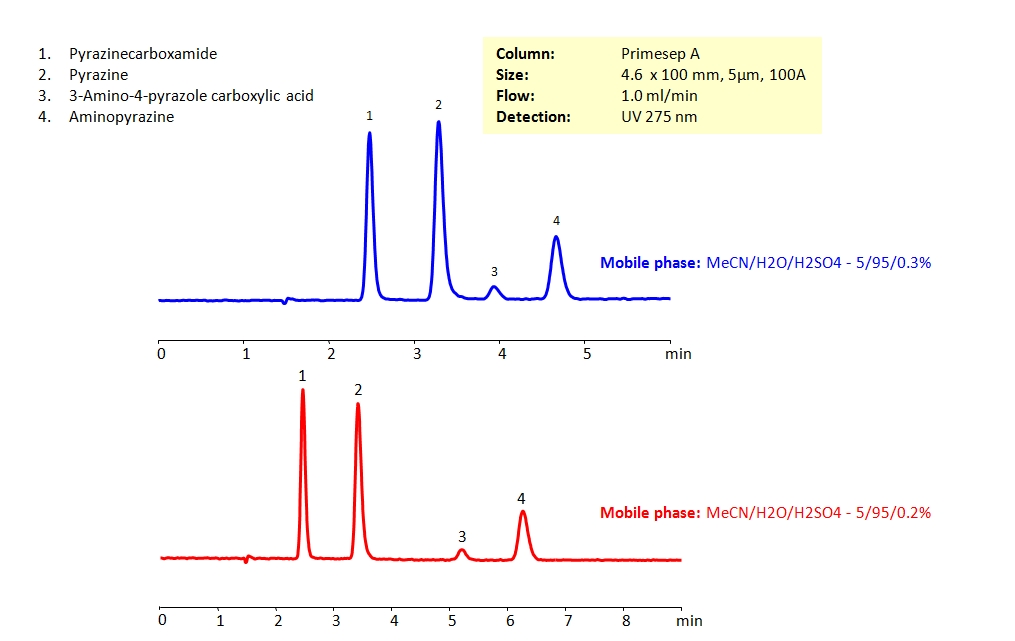
HPLC Separation of Pyrazinecarboxamide and Related Compounds
June 25, 2020
HPLC Method for Pyrazine, 2-Aminopyrazine, Pyrazinamide, 3-Aminopyrazole-4-carboxylic acid on Primesep A by SIELC Technologies
High Performance Liquid Chromatography (HPLC) Method for Analysis of Pyrazine, 2-Aminopyrazine, Pyrazinamide, 3-Aminopyrazole-4-carboxylic acid.
Pyrazinamide is a medication used in the treatment of infectious disease tuberculosis and is on the World Health Organization’s (WHO) list of essential medicines. It has the chemical formula C5H5N3O.
Pyrazine is an organic compound with C₄H₄N₂ chemical formula. It is known for its sweet and nutty aroma, but is usually used as a therapeutic agent in several drugs. Derivatives of it are also said to have anti-inflammatory, anticancer, antibacterial, and antioxidant properties.
Aminopyrazine, also known as 2-aminopyrazine and pyrazinamide, is a Pyrazine derivative with C₄H₅N₃ chemical formula. It is typically used as a building block in chemical synthesis and is found in pharmaceuticals, especially in antiviral drugs and other agrochemicals. Structure wise, it is a pyrazine ring with an attached amine group.
It can be retained and separated from other pyrazine compounds, which are structurally similar and can be difficult to separate in reverse-phase HPLC, by using Primesep A mixed-mode column. Primesep A’s stationary phase is embedded with strong acidic ion-pairing groups. The analytical method is isocratic and uses the mobile phase of acetonitrile (ACN) and water with sulfuric acid (H2SO4) as buffer and UV detection at 275nm.
| Column | Primesep A, 4.6 x 100 mm, 5 µm, 100 A, dual ended |
| Mobile Phase | MeCN/H2O – 5/95% |
| Buffer | H2SO4 |
| Flow Rate | 1.0 ml/min |
| Detection | UV 275 nm |
| Class of Compounds | Hydrophilic, Heterocyclic, Aromatic |
| Analyzing Compounds | Pyrazine, 2-Aminopyrazine, Pyrazinamide, 3-Aminopyrazole-4-carboxylic acid |
Application Column
Primesep A
Column Diameter: 4.6 mm
Column Length: 100 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended
3-Aminopyrazole-4-carboxylic acid
Pyrazinamide
Pyrazine