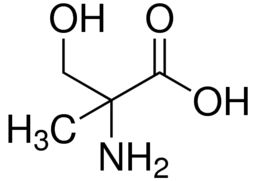
| CAS Number | 2480-26-4 |
|---|---|
| Molecular Formula | C4H9NO3 |
| Molecular Weight | 119.120 |
| InChI Key | PSFABYLDRXJYID-VKHMYHEASA-N |
| LogP | -1.89 |
| Synonyms |
|
Applications:
HPLC Method for Separation of Serine, a-Methylserine, GABA, and Methionine Mixture on Primesep 100 column
November 21, 2010
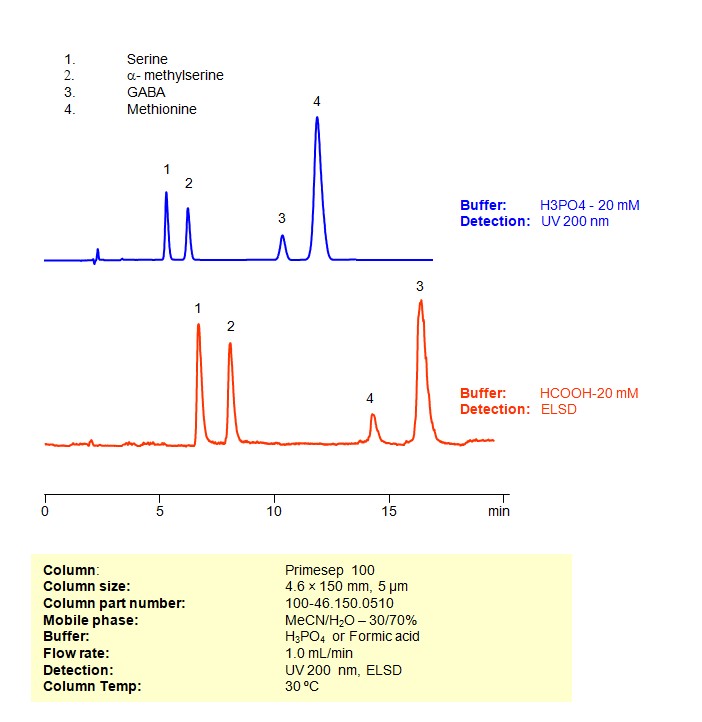
Four underivatized amino acids (serine, methylserine, GABA and methionine) were separated on a Primesep 100 reversed-phase cation-exchange mixed-mode HPLC column. Two methods, one for UV and one for ELSD/LC/MS show good separation and peak shape for underivatized amino acids. This column and general HPLC approach can be used for analysis of underivatized amino acids. Primesep 100 is designed to replace reversed-phase HPLC column in combination with ion-pairing reagents.
| Column | Primesep 100, 4.6×250 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | H3PO4 or Formic acid |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 200 nm, ELSD |
| Class of Compounds | Drug, Acid, Hydrophilic, Ionizable, Vitamin, Supplements, Amino acid |
| Analyzing Compounds | Serine, Methylserine, GABA, Methionine |
Application Column
Primesep 100
Column Diameter: 4.6 mm
Column Length: 150 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended
Serine
a-Methylserine
gamma-Aminobutyric Acid (GABA)

HPLC Separation of Amino Acids
December 6, 2007

Amino acids are building blocks for peptides and proteins. Serine is not essential to human diet and it is synthesized in human body. Methionine is not synthesized in human body and needs to be ingested. GABA is used to enhance growth of specified plants, prevent development of powdery mildew on grapes, and suppress certain other plant diseases. In humans GABA helps to maintain normal brain function. Serine, methylserine, GABA and methionine are separated on Primesep 100 column by mixed-mode mechanism. Amino acids are retained by combination of reverse phase and cation-exchange mechanisms. At lower pH carboxylic acid fragment of amino acid is suppressed and not ionized, making amino acids more basic and slightly more hydrophobic. Amount of acetonitrile, buffer concentration and buffer pH can be used to adjust retention time. Underivatized amino acids are well retained and separated with perfect peak shape and symmetry. Fast method can be used for UV, ESLD and LC/MS quantitation of serine, methylserine, GABA and methionine.
| Column | Primesep 100, 4.6×250 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | H3PO4 |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 200 nm, ELSD |
| Class of Compounds |
Drug, Acid, Hydrophilic, Ionizable, Vitamin, Supplements, Amino acid |
| Analyzing Compounds | Serine, Methylserine, GABA, Methionine |
Application Column
Primesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsSerine
a-Methylserine
gamma-Aminobutyric Acid (GABA)
UV Detection