HPLC Method for Ketoprofen on Legacy L1 by SIELC Technologies
High Performance Liquid Chromatography (HPLC) Method for Analysis of Ketoprofen.
Ketoprofen is a prescription anti-inflammatory drug (NSAID) that is used to treat pain, inflammation, and fever. It is also used in veterinary medicine. It works through inhibiting cyclooxygenase enzymes, which prevents the synthesis of prostaglandins. It can be administered through capsules, topical gel, injections, and patches. It has the chemical formula C16H14O3.
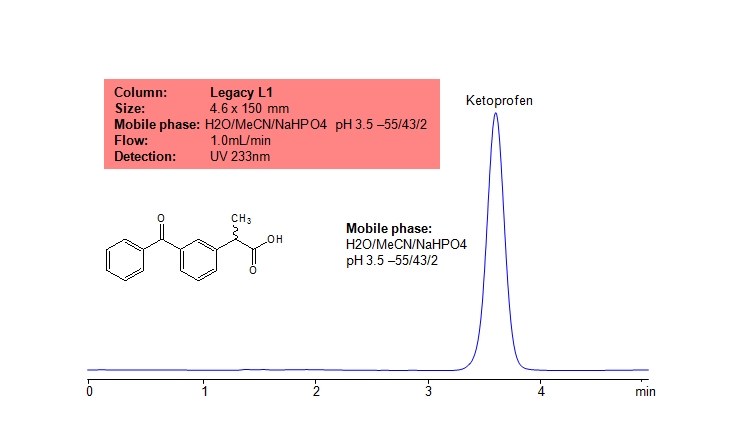
Ketoprofen can be retained and analyzed using the Legacy L1 stationary phase column. The analysis utilizes an isocratic method with a simple mobile phase consisting of water and acetonitrile (MeCN) with a sulfuric acid buffer. Detection is performed using UV.
| Column | Legacy L1, 4.6 x 150 mm, 5 µm, 100 A, dual ended |
| Mobile Phase | MeCN/H2O – 43/55% |
| Buffer | NaHPO4 pH3.5 – 2% |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 233 nm |
| Class of Compounds |
Drug, Acid, Ionizable, Hydrophobic |
| Analyzing Compounds | Ketoprofen |
Application Column
Legacy L1
Column Diameter: 4.6 mm
Column Length: 150 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended