HPLC Method for Aceclidine on Primesep 100 by SIELC Technologies
High Performance Liquid Chromatography (HPLC) Method for Analysis of Aceclidine
Aceclidine historically unapproved in the United States, received regulatory authorization from the U.S. Food and Drug Administration in July 2025 in the form of an ophthalmic solution (VIZZ, aceclidine 1.44%) for the treatment of presbyopia, marking its transition from an experimental cholinergic agent to a clinically utilized therapeutic.
Drug class: Muscarinic cholinergic agonist
Mechanism of action: Stimulates muscarinic receptors, leading to smooth muscle contraction and glandular secretion.
Historical use: Studied for glaucoma, gastrointestinal motility, and other parasympathetic-related effects
You can find detailed UV spectra of Aceclidine and information about its various lambda maxima by visiting the following link.
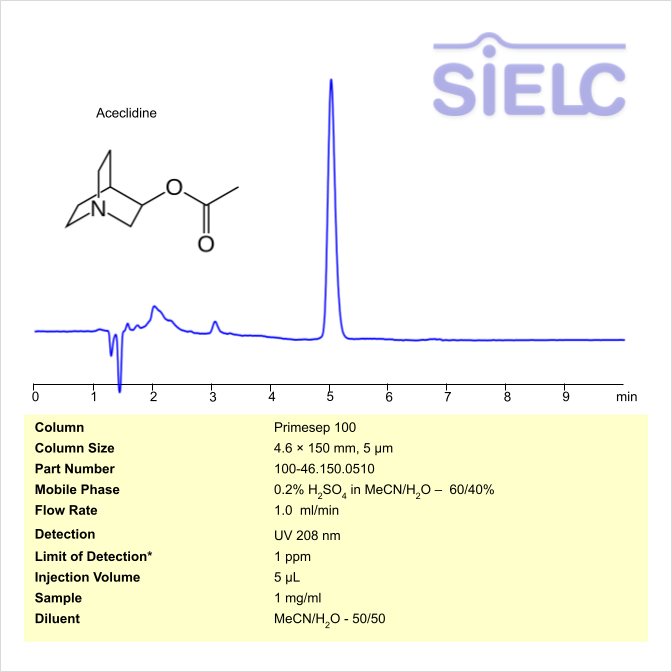
Aceclidine can be retained and analyzed using the Primesep 100 stationary phase column. The analysis utilizes an isocratic method with a simple mobile phase consisting of water and acetonitrile (MeCN) with a sulfuric acid as a buffer. Detection is performed using UV.
| Column | Primesep 100, 4.6 x 150 mm, 5 µm, 100 A, dual ended |
| Mobile Phase | MeCN/H2O – 60/40 % |
| Buffer | H2SO4 – 0.2% |
| Flow Rate | 1.0 ml/min |
| Detection | UV 208 nm |
| Class of Compounds | Drug |
| Analyzing Compounds | Aceclidine |
Application Column
Primesep 100
Column Diameter: 4.6 mm
Column Length: 150 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended