HPLC Method for EDTA (Ethylenediaminetetraacetic Acid) on Newcrom B by SIELC Technologies
High Performance Liquid Chromatography (HPLC) Method for Analysis of EDTA (Ethylenediaminetetraacetic Acid).
EDTA Standards Solution A:
For the preparation of the EDTA standard solution, 5 mg of EDTA was accurately weighed and transferred into a 5 mL volumetric flask and dissolved in 0.001N NaOH water solution with sonication or magnetic stirrer mixing. Filtered The EDTA stock solution (1.0 mg/mL) should be stored in a cold dark place and can be used for a week to prepare standards of required concentration.
Iron(III) chloride Solution B:
The standard stock solution of Iron(III) chloride (10 mg/ml) was prepared in water. 50 mg of FeCl3 was accurately weighed and transferred into a 5 mL volumetric flask and dissolved in water, with sonication if needed.
General procedure for Ferric EDTA complex analysis:
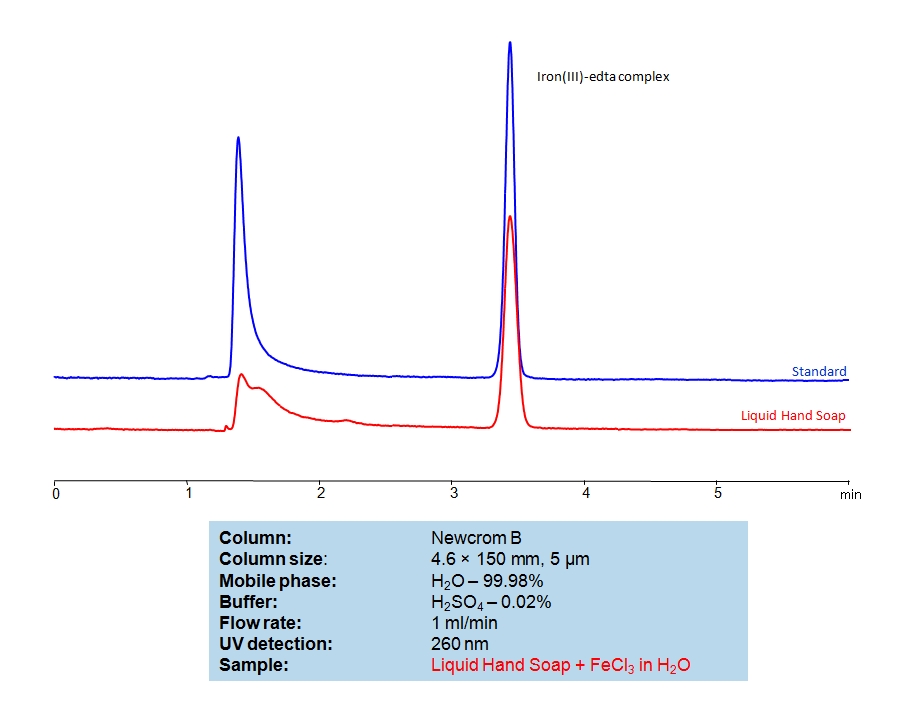
To make a sample for analysis mix 100 µL Solution A (or unknown sample) with 100 µL Solution B and 800 µL of water. Place this mixture in a plastic HPLC vial for analysis. Setup instrument and column according to the method provided.
You can find detailed UV spectra of EDTA (Ethylenediaminetetraacetic Acid) and information about its various lambda maxima by visiting the following link.
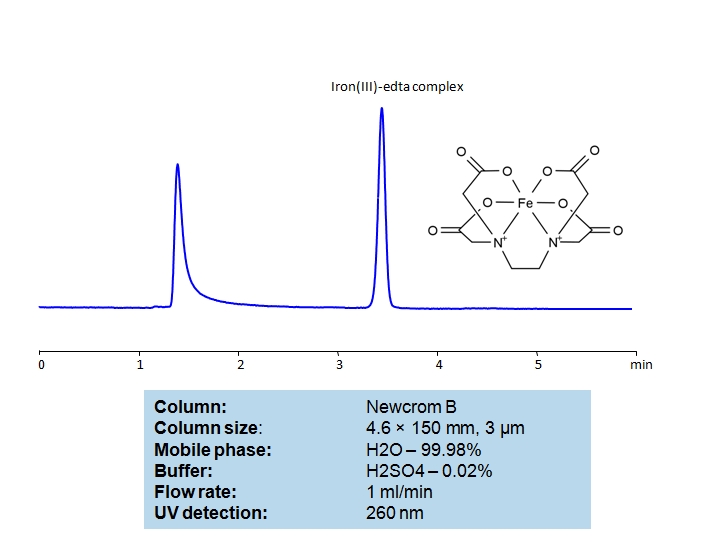
EDTA (Ethylenediaminetetraacetic Acid) can be retained and analyzed using the Newcrom B stationary phase column. The analysis utilizes an isocratic method with a simple mobile phase consisting of water and acetonitrile (MeCN) with a sulfuric acid buffer. Detection is performed using UV.
| Column | Newcrom B, 4.6 x 150 mm, 5 µm, 100 A, dual ended |
| Mobile Phase | H2O – 99.98% |
| Buffer | H2SO4 – 0.02% |
| Flow Rate | 1.0 ml/min |
| Detection | UV 260nm |
| Class of Compounds | Acid, Hydrophilic |
| Analyzing Compounds | EDTA (Ethylenediaminetetraacetic Acid) |
Application Column
Newcrom B
Column Diameter: 4.6 mm
Column Length: 150 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended