| CAS Number | 1563-66-2 |
|---|---|
| Molecular Formula | C12H15NO3 |
| Molecular Weight | 221.256 |
| InChI Key | DUEPRVBVGDRKAG-UHFFFAOYSA-N |
| LogP | 2.32 |
| Synonyms |
|
Applications:
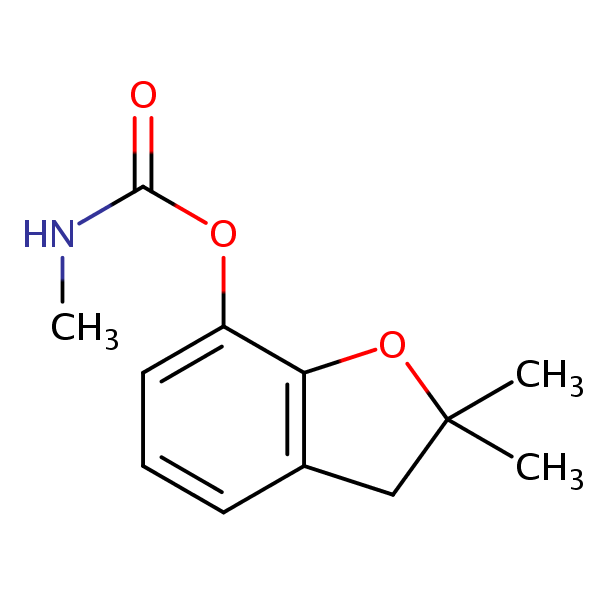
HPLC Separation of Pesticides, Herbicides, Fungicides and Insecticides on Newcrom BH Column
May 22, 2020
Herbicides are used to control unwanted plants, they are also known as weedkillers. Insecticides are used to kill insects. Fungicides are used to kill parasitic fungi. Pesticide is a more generic term that includes herbicides, fungicides and insecticides in its definition. All are heavily used in agriculture. By using HPLC, many different pesticides can be separated and their retention characteristics controlled using the Newcrom BH mixed-mode column. It can be used with different organic media such as acetonitrile (ACN) or methanol (MeOH). By varying the concentration of organic modifier and using different buffers like sulfuric acid (H2SO4), ammonium formate (AmFm), or formic acid, separation of desired pesticides can be achieved.
| Column | Newcrom BH, 3.2×100 mm, 5 µm, 100A |
| Mobile Phase | MeCN, MeOH |
| Buffer | H2SO4, Formic acid, AmFm pH 3.0 |
| Flow Rate | 0.6 ml/min |
| Detection | UV 275 nm |
| Class of Compounds | Pesticides, Herbicides, Fungicides, Insecticides |
| Analyzing Compounds | Monocrotophos, Metribuzin, Bromacil, Carbofuran, 4-Chlorophenoxyacetic acid, Bentazone, Folpet, Fenthion, Fenvalerate, Metrabuzin, Carbofuran, Atrazine, Parathion –methyl, MCPA, Clopyralid, Dicamba, Benthazon, Simazine, Diuron, MCPB, Mecoprop, 4-Chlorophenoxyacetic acid,2,4,5-T |
Application Column
Newcrom BH
The Newcrom columns are a family of reverse-phase-based columns. Newcrom A, AH, B, and BH are all mixed-mode columns with either positive or negative ion-pairing groups attached to either short (25 Å) or long (100 Å) ligand chains. Newcrom R1 is a special reverse-phase column with low silanol activity.
Select options4-CPA
Atrazine
Bentazone-sodium
Carbofuran
Clopyralid
Dicamba
Diuron
Fenvalerate
Folpet
MCPA
MCPB
Mecoprop
Methyl parathion
Metribuzin
Monocrotophos
Simazine

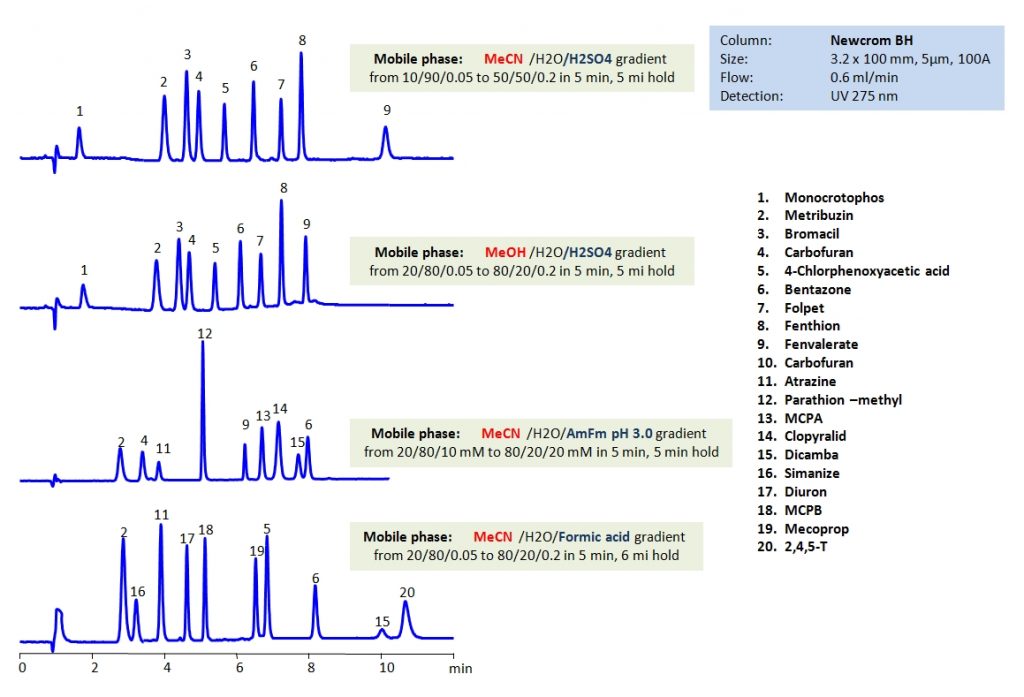
HPLC Separation of Carbofuran on Obelisc R and Primesep 100 Columns
September 14, 2015
Carbofuran is an insecticide notable for its high toxicity to vertebrates, especially birds; it is one of the most toxic to humans as well. While band in many countries such as the U.S. and Canada, it can be purchased over-the-counter in some countries such as Kenya. Carbofuran was considered a target pesticide by the EURL (European Union Reference Laboratory) for the EUPT-FF9 2015 a proficiency test for the analysis of a over a hundred pesticides. Obelisc R and Primesep 100 were used to analyze carbofuran. Primesep 100 separates by reverse-phase and utilizing embedded acidic ion-pairing groups. Obelisc R uses long hydrophobic chains and multiple ion-pairing groups.
| Column | Obelisc R, 2.1×150 mm, 5 µm, 100A |
| Mobile Phase | Gradient MeCN – 25-70%, 15 min |
| Buffer | Gradient AmAc pH 3.0- 30-60 mM, 15 min |
| Flow Rate | 0.4 ml/min |
| Detection | UV, 270 nm |
| Column | Obelisc R, 2.1×150 mm, 5 µm, 100A |
| Mobile Phase | Gradient MeCN – 10-70%, 15 min |
| Buffer | Gradient AmAc pH 3.0- 20-60 mM, 15 min |
| Flow Rate | 0.4 ml/min |
| Detection | UV, 270 nm |
| Column | Primesep 100, 2.1×150 mm, 5 µm, 100A |
| Mobile Phase | Gradient MeCN – 10-70%, 15 min |
| Buffer | Gradient AmAc pH 3.0- 20-60 mM, 15 min |
| Flow Rate | 0.4 ml/min |
| Detection | UV, 270 nm |
| Class of Compounds |
Insecticide, Herbicide, Fungicide, Hydrophobic, Ionizable |
| Analyzing Compounds | Carbofuran |
Application Column
Obelisc R
SIELC has developed the Obelisc™ columns, which are mixed-mode and utilize Liquid Separation Cell technology (LiSC™). These cost-effective columns are the first of their kind to be commercially available and can replace multiple HPLC columns, including reversed-phase (RP), AQ-type reversed-phase, polar-embedded group RP columns, normal-phase, cation-exchange, anion-exchange, ion-exclusion, and HILIC (Hydrophilic Interaction Liquid Chromatography) columns. By controlling just three orthogonal method parameters - buffer concentration, buffer pH, and organic modifier concentration - users can adjust the column properties with pinpoint precision to separate complex mixtures.
Select optionsPrimesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select options
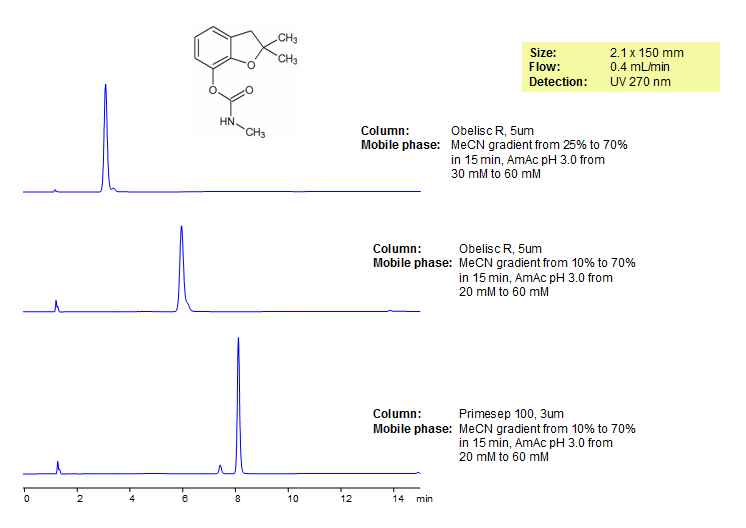
Carbofuran Separation on Obelisc R HPLC Column
August 24, 2015
| Column | Obelisc R, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN – 20% |
| Buffer | AmFm pH 3.0- 20 mM |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 250 nm |
| Class of Compounds |
Insecticide, Herbicide, Fungicide, Hydrophobic, Ionizable |
| Analyzing Compounds | Carbofuran |
Application Column
Obelisc R
SIELC has developed the Obelisc™ columns, which are mixed-mode and utilize Liquid Separation Cell technology (LiSC™). These cost-effective columns are the first of their kind to be commercially available and can replace multiple HPLC columns, including reversed-phase (RP), AQ-type reversed-phase, polar-embedded group RP columns, normal-phase, cation-exchange, anion-exchange, ion-exclusion, and HILIC (Hydrophilic Interaction Liquid Chromatography) columns. By controlling just three orthogonal method parameters - buffer concentration, buffer pH, and organic modifier concentration - users can adjust the column properties with pinpoint precision to separate complex mixtures.
Select options
Carbofuran Separation on Primesep and Obelisc Mixed-Mode HPLC Columns
August 24, 2015
Separation type: Liquid Chromatography Mixed-mode
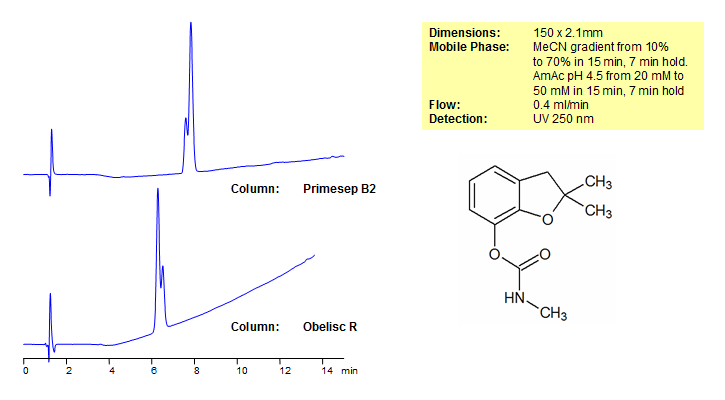
Carbofuran is one of the most acutely toxic insecticides used on field crops. It is banned in Europe, Canada, and as of 2009 was banned in the United States. The EURL (European Union Reference Laboratory), included carbofuran as a target pesticide for the EUPT-CF9 a proficiency test for cereals and feedingstuff that require muli-residue methods. Primesep B2 and Obelisc R were used to retain carbofuran and separate it from impurities. Primesep B2 contains embedded basic ion-pairing groups and Obelisc R contains embedded ionic and hydrophobic groups which can assist in fine tuning separations. Method is LC/MS compatible and can be used as a general approach for analyzing carbofuran and other phenoxy acids.
| Column | Primesep B2, 2.1×150 mm, 5 µm, 100A |
| Mobile Phase | Gradient MeCN – 10-70%, 15 min, 7 min hold |
| Buffer | Gradient AmAc pH 4,5- 20-50 mM, 15 min, 7 min hold |
| Flow Rate | 0.4 ml/min |
| Detection | UV, 250 nm |
| Column | Obelisc R, 2.1×150 mm, 5 µm, 100A |
| Mobile Phase | Gradient MeCN – 10-70%, 15 min, 7 min hold |
| Buffer | Gradient AmAc pH 4,5- 20-50 mM, 15 min, 7 min hold |
| Flow Rate | 0.4 ml/min |
| Detection | UV, 250 nm |
| Class of Compounds |
Insecticide, Herbicide, Fungicide, Hydrophobic, Ionizable |
| Analyzing Compounds | Carbofuran |
Application Column
Obelisc R
SIELC has developed the Obelisc™ columns, which are mixed-mode and utilize Liquid Separation Cell technology (LiSC™). These cost-effective columns are the first of their kind to be commercially available and can replace multiple HPLC columns, including reversed-phase (RP), AQ-type reversed-phase, polar-embedded group RP columns, normal-phase, cation-exchange, anion-exchange, ion-exclusion, and HILIC (Hydrophilic Interaction Liquid Chromatography) columns. By controlling just three orthogonal method parameters - buffer concentration, buffer pH, and organic modifier concentration - users can adjust the column properties with pinpoint precision to separate complex mixtures.
Select optionsPrimesep B2
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select options