| CAS Number | 506-32-1 |
|---|---|
| Molecular Formula | C20H32O2 |
| Molecular Weight | 304.474 |
| InChI Key | YZXBAPSDXZZRGB-DOFZRALJSA-N |
| LogP | 6.56 |
| Synonyms |
|
Applications:
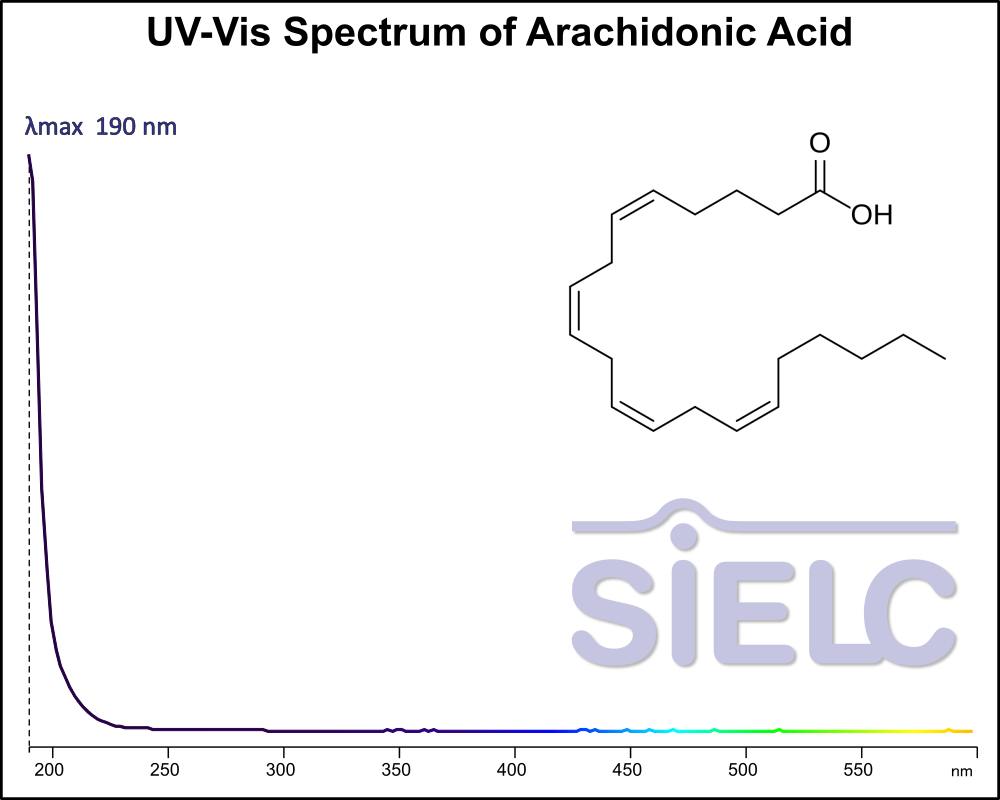
Uv-Vis Spectrum of Arachidonic acid
March 3, 2026
Access the UV-Vis Spectrum SIELC Library
If you are looking for optimized HPLC method to analyze Arachidonic acid check our HPLC Applications library
For optimal results in HPLC analysis, it is recommended to measure absorbance at a wavelength that matches the absorption maximum of the compound(s) being analyzed. The UV spectrum shown can assist in selecting an appropriate wavelength for your analysis. Please note that certain mobile phases and buffers may block wavelengths below 230 nm, rendering absorbance measurement at these wavelengths ineffective. If detection below 230 nm is required, it is recommended to use acetonitrile and water as low UV-transparent mobile phases, with phosphoric acid and its salts, sulfuric acid, and TFA as buffers.
For some compounds, the UV-Vis Spectrum is affected by the pH of the mobile phase. The spectra presented here are measured with an acidic mobile phase that has a pH of 3 or lower.

HPLC ELSD Method for Analysis of Arachidonoyl ethanolamideand Arachidonic acid on Lipak Column
December 18, 2024
HPLC Method for , Arachidonoyl ethanolamide (AEA), Arachidonic acid on Lipak by SIELC Technologies
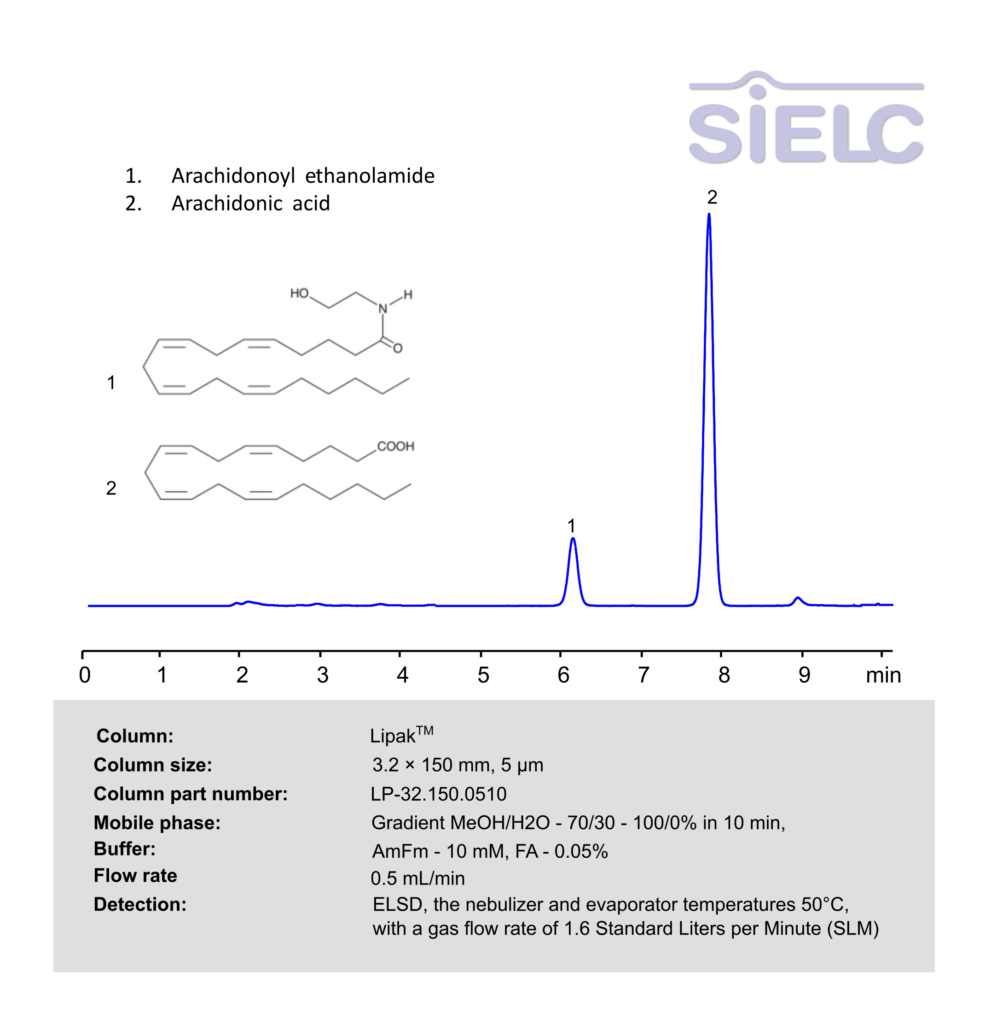
High Performance Liquid Chromatography (HPLC) Method for Analysis of , Arachidonoyl ethanolamide (AEA), Arachidonic acid
Arachidonoyl ethanolamide (AEA) and arachidonic acid (AA) are closely related bioactive lipids that play significant roles in various physiological and biochemical processes.
Arachidonic acid, a polyunsaturated omega-6 fatty acid, is a critical precursor for the biosynthesis of prostaglandins, thromboxanes, and leukotrienes. In cells, nearly all arachidonic acid is stored as an esterified component of membrane phospholipids, with its levels tightly regulated through interconnected metabolic pathways. Upon stimulation, free arachidonic acid is transiently released, serving as a vital substrate for the production of eicosanoid signaling molecules. Processes such as receptor-mediated release, metabolic transformation, and reuptake of free arachidonate are essential for cell signaling and inflammatory responses.
Precursor Relationship: AEA is synthesized from arachidonic acid via enzymatic pathways. AA is first converted to N-arachidonoyl phosphatidylethanolamine (NAPE), then cleaved to form AEA.Biological Cross-Talk:
- Both AEA and AA influence inflammatory and pain pathways.
- AEA’s degradation contributes to free arachidonic acid pools, linking endocannabinoid signaling with eicosanoid production.
, Arachidonoyl ethanolamide (AEA), Arachidonic acid can be retained, and analyzed using a Lipak mixed-mode stationary phase column. The analysis utilizes an gradient method with a mobile phase consisting of water, methanol (MeOH), ammonium formate and formic acid as a buffer. Detection is achieved using ELSD
| Column | Lipak, 3.2 x 150 mm, 5 µm, 100 A, dual ended |
| Mobile Phase | Gradient MeOH/H2O – 70/30 – 100/0% in 10 min |
| Buffer | AmFm– 10 mM, FA – 0.05% |
| Flow Rate | 0.5 ml/min |
| Detection | ELSD, the nebulizer and evaporator temperatures 50°C, with a gas flow rate of 1.6 Standard Liters per Minute (SLM) |
| Class of Compounds | Endocannabinoids |
| Analyzing Compounds | , Arachidonoyl ethanolamide (AEA), Arachidonic acid |
Application Column
Lipak
Column Diameter: 3.2 mm
Column Length: 150 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended
Arachidonic acid
Arachidonoyl ethanolamide (AEA)

Separation of Arachidonic acid on Newcrom R1 HPLC column
February 16, 2018
Arachidonic acid can be analyzed by this reverse phase (RP) HPLC method with simple conditions. The mobile phase contains an acetonitrile (MeCN), water, and phosphoric acid. For Mass-Spec (MS) compatible applications the phosphoric acid needs to be replaced with formic acid. Smaller 3 µm particles columns available for fast UPLC applications. This liquid chromatography method is scalable and can be used for isolation impurities in preparative separation. It also suitable for pharmacokinetics.
Application Column
Newcrom R1
The Newcrom columns are a family of reverse-phase-based columns. Newcrom A, AH, B, and BH are all mixed-mode columns with either positive or negative ion-pairing groups attached to either short (25 Å) or long (100 Å) ligand chains. Newcrom R1 is a special reverse-phase column with low silanol activity.
Select options



