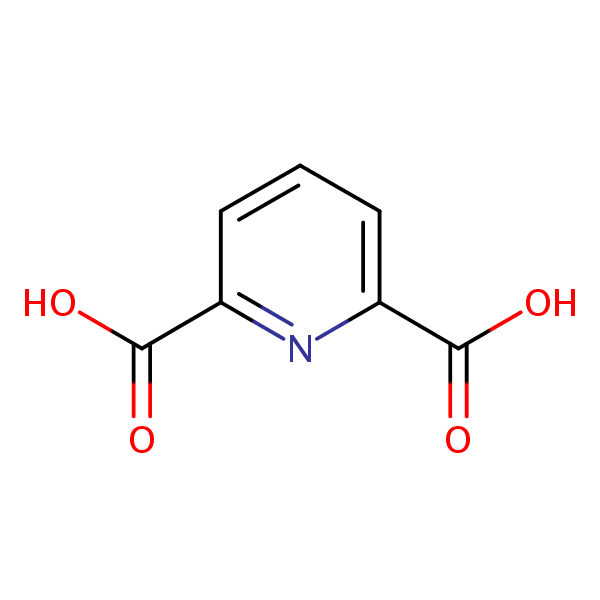
| CAS Number | 499-83-2 |
|---|---|
| Molecular Formula | C7H5NO4 |
| Molecular Weight | 167.120 |
| InChI Key | WJJMNDUMQPNECX-UHFFFAOYSA-N |
| LogP | 0.0746 |
| Synonyms |
|
Applications:
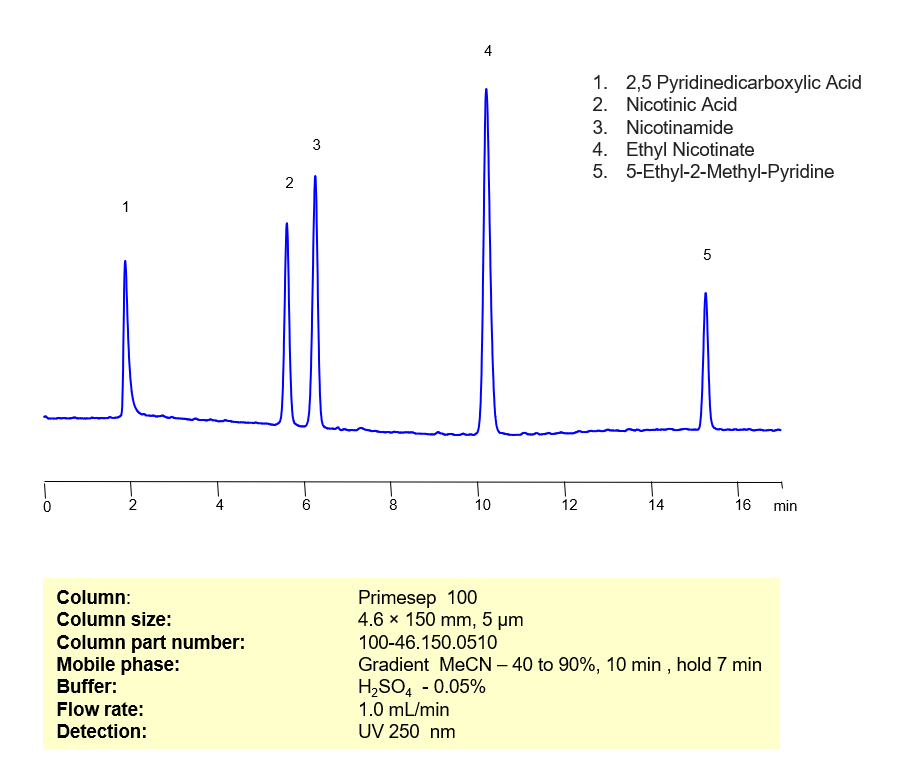
HPLC Method for Simultaneous Determination of Nicotinic Acid, Nicotinamide, Pyridine Dicarboxylic Acid, Ethyl Methyl Pyridine and Ethyl Nicotinate on Primesep 100 Column
August 29, 2022
Separation type: Liquid Chromatography Mixed-mode
High Performance Liquid Chromatography (HPLC) Method for Analysis of Nicotinic Acid, Nicotinamide, Pyridine Dicarboxylic Acid, Ethyl Methyl Pyridine and Ethyl Nicotinate Nicotinic acid, also known as Niacin or Vitamin B3, is an essential nutrient that plants and animals synthesize from tryptophan. It is also used as a dietary supplement to treat pellagra. A derivative of Nicotinic acid, Nictotinamide, is a key component of the coenzymes nicotinamide adenine dinucleotide (NAD) and nicotinamide adenine dinucleotide phosphate (NADP+). Pyridine Dicarboxylic acid (PDA), or Dipicolinic acid (DPA), is a key factor in bacterial endospore heat resistance. It also plays an important role in preparing transition metal complexes for ion chromatography. Ethyl Methyl Pyridine is a common precursor to Nicotinic acid. Ethyl nicotinate, or nicotine acid ethyl ester, is a popular topical treatment for ligament, joint, and muscle pain. These 5 organic compounds can be separated, retained, and analyzed on a Primesep 100 mixed-mode column using an analytical method with a simple gradient mobile phase of water, Acetonitrile (MeCN), and a sulfuric acid (H2SO4) buffer. This analysis method can be UV detected at 250 nm with high resolution and peak symmetry.
| Column | Primesep 100, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN – Gradient |
| Buffer | H2SO4 – 0.05% |
| Flow Rate | 1.0 ml/min |
| Detection | UV 250 nm |
| Class of Compounds | Drug, Acid |
| Analyzing Compounds | Nicotinic Acid, Nicotinamide, Pyridine Dicarboxylic Acid, Ethyl Methyl Pyridine and Ethyl Nicotinate |
Application Column
Primesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select options5-Ethyl-2-methylpyridine
Nicotinamide
Nicotinic Acid/Niacin (3-pyridinecarboxylic acid)
Pyridinedicarboxylic Acid

HPLC Separation of Pyridinecarboxylic Acids
March 27, 2011
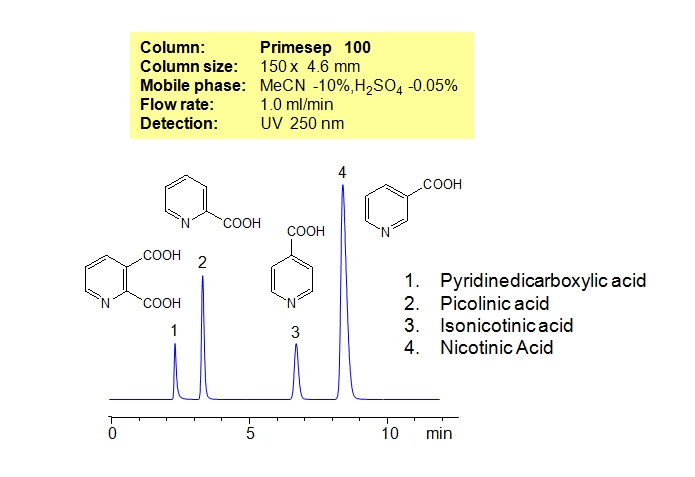
Pyridinecarboxylic acids exist as three isomers with different position of carboxylic acid relative to nitrogen in pyridine. Three isomers of pyridinecarboxylic acid (picolinic or 2-pyridinecarvoxylic acid, niacin or 3-pyridinecarboxylic acid, isonicotinic or 4-pyridinecarboxylic acid), along with pyridinedicarboxylic acid, are separated on a Primesep 100 column. Pyridinecarboxylic acids have a similar empirical formula, and are very similar in terms of hydrophobicity and ionic properties. Small differences in these properties are enough to achieve good separation on cation-exchange mixed-mode HPLC column like Primesep 100. Retention time for all compounds is controlled by the amount of acetonitrile and amount of ions in the mobile phase. Ions in the mobile phase can be created by organic and inorganic acids and corresponding salt buffers. Various detection techniques can be used for monitoring pyridinecarboxylic acids. Other ionizable isomers can be successfully separated on mixed-mode columns.
Application Column
Primesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsNicotinic Acid/Niacin (3-pyridinecarboxylic acid)
Organic Acids
Picolinic Acid
Pyridinedicarboxylic Acid