Separation type: Bridge Ion Separation Technology, or BIST™ by SIELC Technologies
HPLC Method for Analysis of Sucrose octasulfate on BIST A+ Column by SIELC Technologies
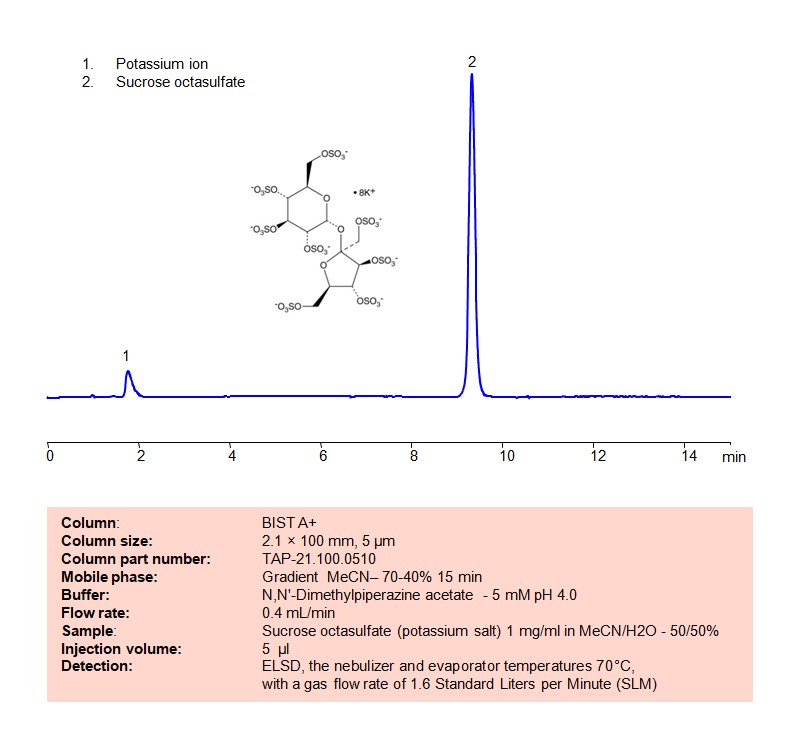
High Performance Liquid Chromatography (HPLC) Method for Analysis of Sucrose octasulfate
Sucrose octasulfate is a chemical compound derived from sucrose, a common sugar. It is characterized by the substitution of eight sulfate groups for hydroxyl groups in the sucrose molecule. This modification significantly changes the properties of the original sugar molecule, making sucrose octasulfate more complex and specialized in its applications.
In terms of its structure, sucrose is a disaccharide composed of glucose and fructose units. When these hydroxyl groups are replaced with sulfate groups in sucrose octasulfate, the molecule becomes highly polar and acidic due to the presence of sulfate groups, which are strong acid residues.
Using SIELC’s newly introduced BIST™ method, sucrose octasulfate can be retained on a negatively-charged, cation-exchange BIST™ A column. There are two keys to this retention method: 1) a multi-charged, positive buffer, such as TMEDA formate, which acts as a bridge, linking the negatively charged dye to the negatively-charged column surface and 2) a mobile phase consisting mostly of organic solvent (such as MeCN) to minimize the formation of a solvation layer around the charged analytes. Using this new and unique analysis method, oligonucleotide can be separated, retained, and detected at ELSD
Please read more on oligonucleotides analysis by HPLC in our April’s 2023 newsletter.
Condition
| Column | BIST A+, 2.1 x 100 mm, 5 µm, 100 A, dual ended |
| Mobile Phase | Gradienr MeCN – 70-40%, 15 min |
| Buffer | DMP acetate pH 4.0 – 5 mM |
| Flow Rate | 0.4 ml/min |
| Detection | ELSD, the nebulizer and evaporator temperatures 70°C, with a gas flow rate of 1.6 Standard Liters per Minute (SLM |
| Sample | 1 mg/ml in MeCN/H2O – 50/50% |
| Injection volume | 5 µl |
| LOD* |
Description
| Class of Compounds | Sulfated polysaccharides |
| Analyzing Compounds | Sucrose octasulfate |
Application Column
BIST A+
Column Diameter: 2.1 mm
Column Length: 100 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended





