HPLC Method for Analysis of (Compound not found) on (Column not found) by SIELC Technologies
High Performance Liquid Chromatography (HPLC) Method for Analysis of (Compound not found)
Procainamide and procaine are two different pharmaceutical compounds with distinct uses and properties.
Procainamide:
- Class: Procainamide is an antiarrhythmic medication, belonging to the Class Ia antiarrhythmic drugs. It is used to treat certain types of abnormal heart rhythms (arrhythmias) by stabilizing the cell membrane and reducing excitability in the heart muscle.
- Mechanism of Action: Procainamide works by inhibiting sodium channels in cardiac cells, prolonging the action potential duration and refractory period, which helps in controlling irregular heartbeats.
- Administration: It is typically administered intravenously or orally under medical supervision.
- Medical Use: Procainamide is prescribed for conditions such as atrial and ventricular arrhythmias.
Procaine:
- Class: Procaine is a local anesthetic drug, belonging to the amino ester group of local anesthetics.
- Mechanism of Action: Procaine works by blocking nerve impulses and causing temporary loss of sensation in a specific area of the body.
- Administration: It is usually administered through injection to induce local anesthesia. Procaine is commonly used for minor surgical procedures and dental work.
- Medical Use: Procaine is employed for local anesthesia, providing pain relief in various medical and dental settings.
Procainamide is primarily used for cardiac conditions, while procaine is utilized as a local anesthetic for pain management during medical and dental procedures. While both compounds have a shared ancestry in PABA and contain amine groups, the specific functional groups (amide vs. ester) and their positions in the molecules contribute to their distinct pharmacological properties. It’s a good observation that small structural changes can lead to significant differences in the pharmacological actions of compounds.
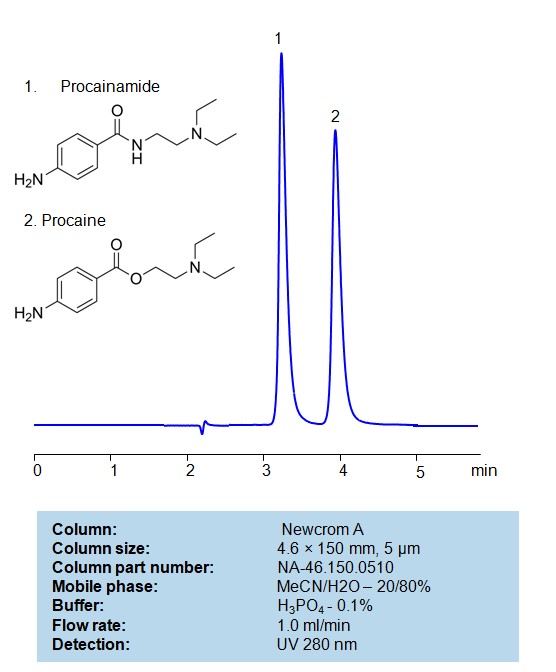
Procainamide and Procaine can be retained, separated and analyzed using a Newcrom A mixed-mode stationary phase column. The analysis utilizes an isocratic method with a simple mobile phase consisting of water, acetonitrile (MeCN), and phosphoricacid as a buffer. Detection is achieved using UV 280 nm
| Column | (Column variation not found) |
| Mobile Phase | MeCN/H2O -20/80% |
| Buffer | H3PO4 – 0.1% |
| Flow Rate | 1.0 ml/min |
| Detection | UV 280 nm |
| Class of Compounds | Drug |
| Analyzing Compounds | (Compound not found) |
