High Performance Liquid Chromatography (HPLC) Method for Analysis of Arbidol (Umifenovir) on Primesep 100 by SIELC Technologies.
Separation type: Liquid Chromatography Mixed-mode
Umifenovir, commonly known by its trade name Arbidol, is an antiviral drug. Originally developed in Russia, it has been used primarily in Russia and China for the prophylaxis and treatment of influenza and other respiratory viral infections.
Mechanism of Action:
Umifenovir interferes with the viral ability to penetrate into host cells. It acts by inhibiting the fusion of the viral lipid membrane with the host cell membrane, thus preventing the virus from entering and infecting the cell.
Uses:
- Influenza: Umifenovir has been prescribed for influenza A and influenza B treatment.
- Other Viral Infections: Besides influenza, the drug has also been indicated for prophylaxis and treatment of acute respiratory viral infections (ARVI) and recurrent respiratory infections.
- Potential Role in COVID-19: In the wake of the COVID-19 pandemic, Umifenovir gained attention as a potential treatment for the virus. Some initial studies and clinical trials have been conducted to assess its efficacy against SARS-CoV-2. While it has been used in some countries as a part of COVID-19 treatment protocols, its efficacy specifically against COVID-19 is not firmly established, and research is ongoing.
Side Effects:
Though generally considered to be safe, some reported side effects of Umifenovir include digestive disturbances, allergic reactions, or elevated heart rate. However, side effects are generally rare. As always, any medication’s potential side effects should be discussed with a healthcare provider.
Availability:
While Umifenovir is available in countries like Russia and China, its acceptance and usage differ worldwide. For instance, it hasn’t been approved for antiviral use in the United States.
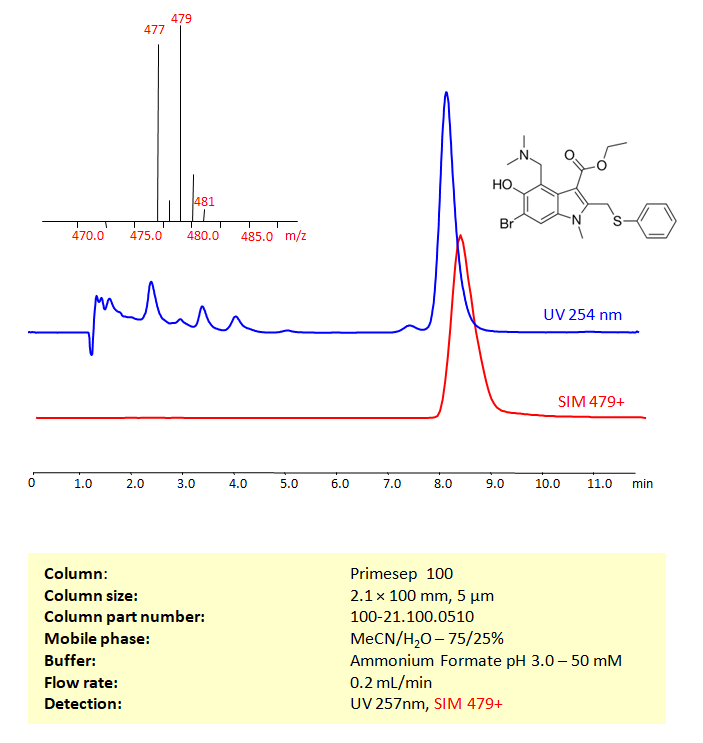
Arbidol can be retained, and analyzed on a Primesep 100 mixed-mode stationary phase column using an isocratic analytical method with a simple mobile phase of water, Acetonitrile (MeCN), and a Ammonium formate as a buffer. This analysis method can be detected using UV at 254 nm, an Evaporative Light Scattering Detector (ELSD), or any other evaporative detection method (CAD, ESI-MS)
High Performance Liquid Chromatography (HPLC) Method for Analyses of Arbidol (Umifenovir)
Condition
| Column | Primesep 100, 2.1 x 100 mm, 5 µm, 100 A, dual ended |
| Mobile Phase | MeCN/H2O – 75/25% |
| Buffer | Ammonium Formate pH 3.0- 50 mM |
| Flow Rate | 0.2 ml/min |
| Detection | UV 254 nm, LCMS SIM 479+ |
| Peak Retention Time | 8.17 min |
Description
| Class of Compounds | Drug |
| Analyzing Compounds | Arbidol (Umifenovir) |
Application Column
Primesep 100
Column Diameter: 2.1 mm
Column Length: 100 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended





