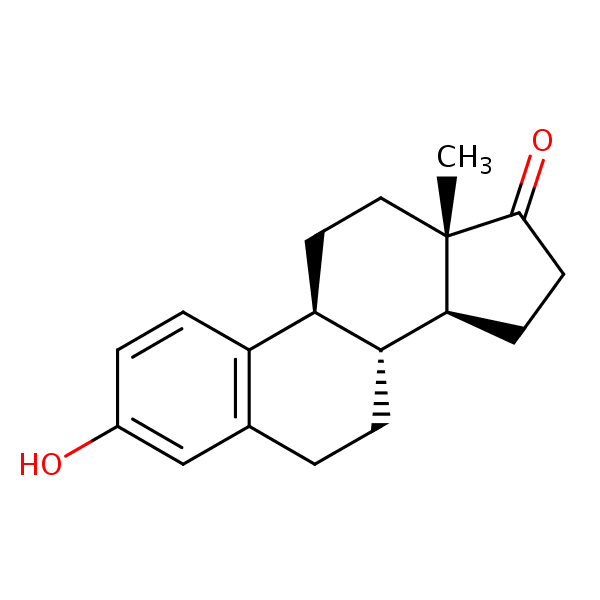
| CAS Number | 53-16-7 |
|---|---|
| Molecular Formula | C18H22O2 |
| Molecular Weight | 270.373 |
| InChI Key | DNXHEGUUPJUMQT-CBZIJGRNSA-N |
| LogP | 3.13 |
| Synonyms |
|
Applications:
HPLC Separation of Estrone
September 9, 2015
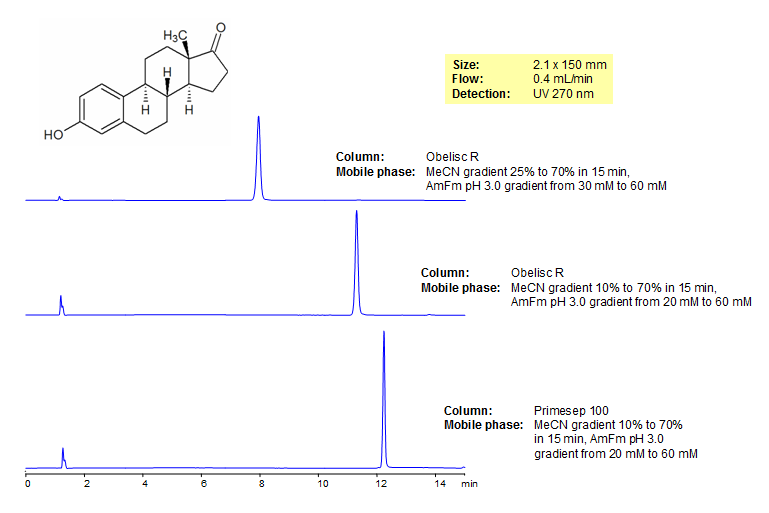
Estrone, also known at oestrone and E1 is a naturally occurring estrogen which is the predominant estrogen in postmenopausal women. It can be found in its long-lived form estrone sulfate. In certain scenarios estrone can be a carcinogen and causes breast pain, nausea, headaches and leg cramps. It was separated on both Obelisc R and Primesep 100 which have unique modes of retention. Method is LC/MS compatible and useful for separating a number of pesticides.
| Column | Obelisc R, 2.1×150 mm, 5 µm, 100A |
| Mobile Phase | Gradient MeCN – 25-70%, 15 min |
| Buffer | Gradient AmAc pH 3.0- 30-60 mM, 15 min |
| Flow Rate | 0.4 ml/min |
| Detection | UV, 270 nm |
| Column | Obelisc R, 2.1×150 mm, 5 µm, 100A |
| Mobile Phase | Gradient MeCN – 10-70%, 15 min |
| Buffer | Gradient AmAc pH 3.0- 20-60 mM, 15 min |
| Flow Rate | 0.4 ml/min |
| Detection | UV, 270 nm |
| Column | Primesep 100, 2.1×150 mm, 5 µm, 100A |
| Mobile Phase | Gradient MeCN – 10-70%, 15 min |
| Buffer | Gradient AmAc pH 3.0- 20-60 mM, 15 min |
| Flow Rate | 0.4 ml/min |
| Detection | UV, 270 nm |
| Class of Compounds |
Hormone, Hydrophobic, Ionizable |
| Analyzing Compounds | Estrone |
Application Column
Obelisc R
SIELC has developed the Obelisc™ columns, which are mixed-mode and utilize Liquid Separation Cell technology (LiSC™). These cost-effective columns are the first of their kind to be commercially available and can replace multiple HPLC columns, including reversed-phase (RP), AQ-type reversed-phase, polar-embedded group RP columns, normal-phase, cation-exchange, anion-exchange, ion-exclusion, and HILIC (Hydrophilic Interaction Liquid Chromatography) columns. By controlling just three orthogonal method parameters - buffer concentration, buffer pH, and organic modifier concentration - users can adjust the column properties with pinpoint precision to separate complex mixtures.
Select optionsPrimesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select options