| CAS Number | 489-46-3 |
|---|---|
| Molecular Formula | C11H19NO9 |
| Molecular Weight | 309.271 |
| InChI Key | SQVRNKJHWKZAKO-UHFFFAOYSA-N |
| LogP | -3.5 |
| Synonyms |
|
Applications:
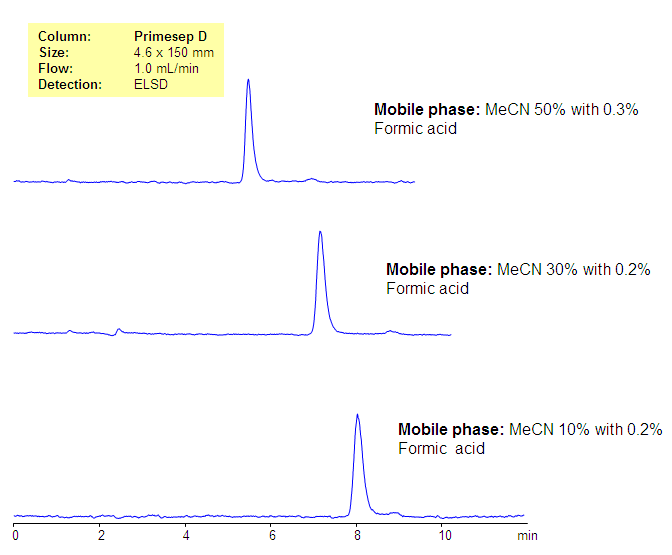
Retention of Sialic Acid on Primesep D Column
July 3, 2013
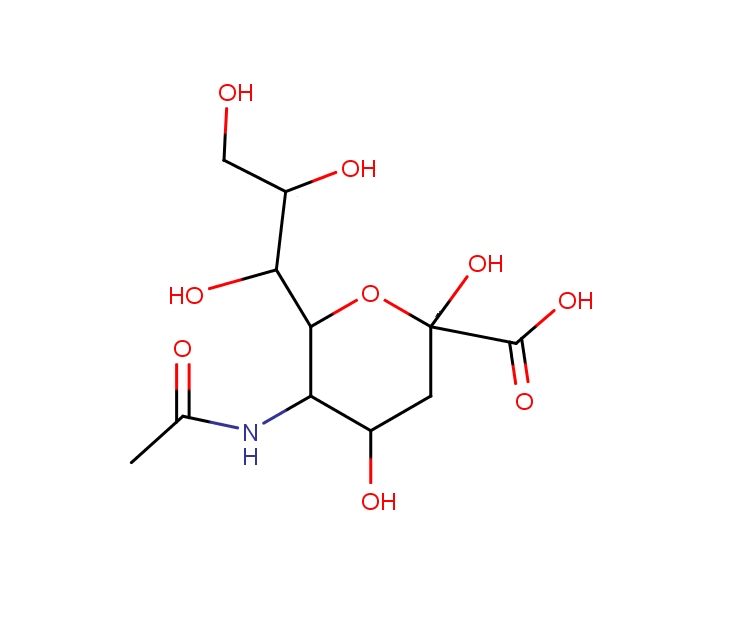
Sialic acid is a nitrogen or oxygen substituted neuraminic acid. It is a sugar-based derivative with very polar properties. The N-substituted compound is acidic in nature. Sialic acid has no retention in reversed-phase, unless an ion-pairing reagent is used. The method for analysis of sialic acid was developed on the Primesep D reversed-phase anion-exchange column. The method is compatible with ELSD and LC/MS and also can be used for the analysis of sialic acid in biological fluids like blood, serum, urine, etc. Retention time of sialic acid is controlled by the pH of the mobile phase and the buffer concentration. It is retained by very weak, reversed-phase and weak, anion-exchange mechanisms.
| Column | Primesep D, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | Formic Acid |
| Flow Rate | 1.0 ml/min |
| Detection | ELSD |
| Class of Compounds |
Drug, Acid, Hydrophilic, Ionizable, Vitamin, Supplements |
| Analyzing Compounds | Sialic Acid |
Application Column
Primesep D
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select options