| CAS Number | 148-24-3 |
|---|---|
| Molecular Formula | C9H7NO |
| Molecular Weight | 145.161 |
| InChI Key | MCJGNVYPOGVAJF-UHFFFAOYSA-N |
| LogP | 2.02 |
| Synonyms |
|
Applications:
HPLC Method for Analysis of 8-hydroxyquinoline
April 30, 2018
Separation type: Liquid Chromatography Mixed-mode
High Performance Liquid Chromatography (HPLC) Method for Analysis of 8-hydroxyquinoline
8-Hydroxyquinoline, or oxine, is a common chelating agent that also serves as an effective antiseptic and antifungal compound. Since it is an effective chelating agent, it is often used in LC applications to determine metal contamination in silica-based columns. Even small amounts of metal ions on the silica surface can cause significant peak distortion with very low peak efficiency and poor peak symmetry.
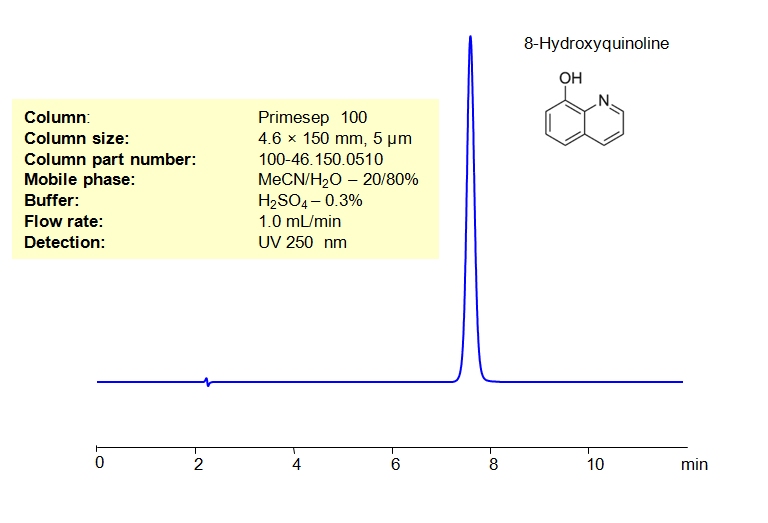
8-Hydroxyquinoline can be retained and analyzed on either a Primesep 100 mixed-mode column using an isocratic analytical method with a simple mobile phase of water, Acetonitrile (MeCN), and a Sulfuric acid (H2SO4) buffer. Compared to a standard C19 column, Primesep 100 offers significantly better retention characteristics due to the ionic interaction of basic quinoline molecules with the cation-exchange functional groups of the column’s stationary phase.
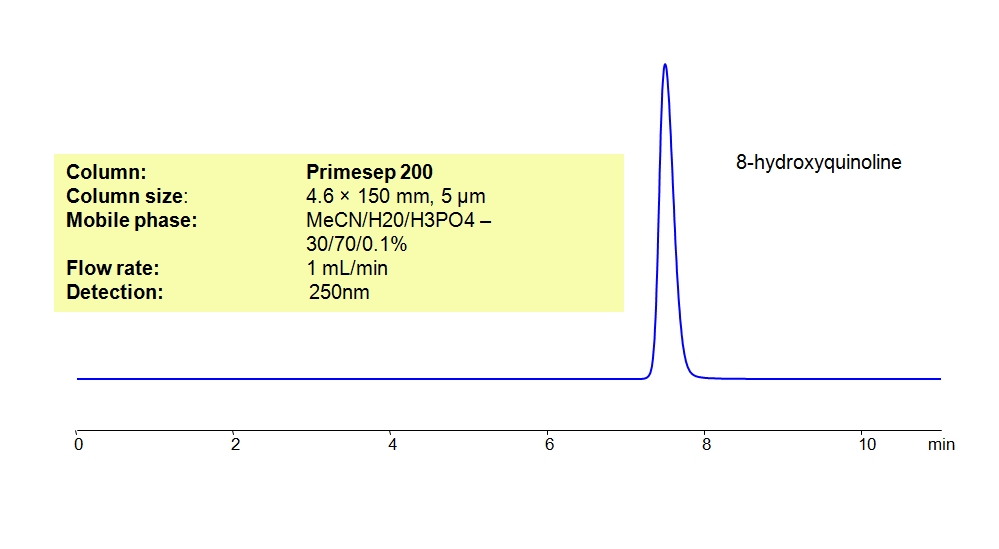
This analytical method can be adapted to a Primesep 200 mixed-mode column by replacing the H2SO4 buffer with Phosphoric acid (H3PO4). The analytical method on either column can be UV detected at 200 nm with high resolution and peak symmetry.
For LC-MS applications, the H3PO4 can be substituted with Ammonium Formate (AmFm) with similar results on a Primesep 200 column.
Primesep 100 also offers much more retention compare to any C18 column due to ionic interaction of basic quinoline molecule with cation-exchange functional groups of the Primesep 100 stationary phase.
Application Column
Primesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsPrimesep 200
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select options