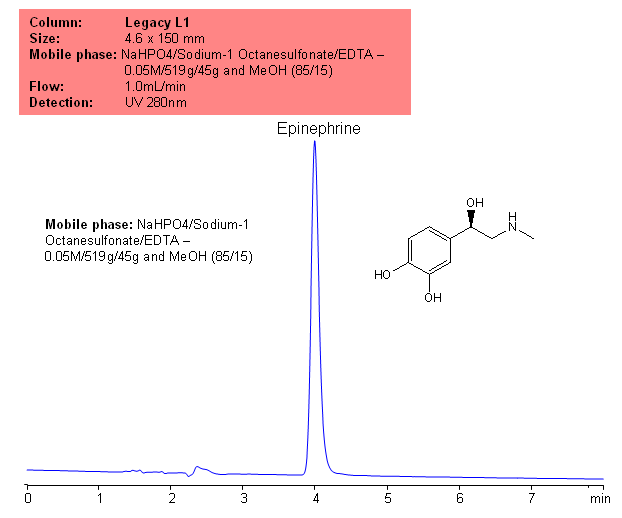
Epinephrine was analyzed according to the US Pharmacopeia method on traditional reversed-phase HPLC columns, classified as a L1 type of column according to USP classification. Legacy L1 column can be used for the analysis of drug substances in various formulations. Legacy L1 can be used in new HPLC methods as well as in validated methods which require experiments based on USP manuscript. The mobile phase can be modified to accommodate other detection techniques, like LC/MS.
| Column | Legacy L1, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeOH – 15% |
| Buffer | Sodium 1 -Octane sulfonate/EDTA |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 280 nm |
| Class of Compounds |
Drug, Neurotransmitter, Hydrophobic, Ionizable |
| Analyzing Compounds | Epinephrine |
Application Column
Legacy L1
SIELC's family of Legacy columns is based on the United States Pharmacopeia's (USP) published chromatographic methods and procedures. Numerous brands have columns used in USP reference standards and methods. USP has created various designations to group together columns with similar types of packing and properties in the solid phase. SIELC's Legacy columns adhere to these strict requirements and properties, allowing you to easily replace older columns that are no longer available without needing to significantly modify your method or SOPs.
Select options




